Advertisements
Advertisements
प्रश्न
Write the name and symbol of the element from the description.
The atom having the smallest size.
Write the name of the atom having the smallest size.
उत्तर
Helium (He)
संबंधित प्रश्न
Answer the following in respect of element `31/15 P`
Give its electronic configuration
Why is the size of neon greater than fluorine?
Which is greater in size an atom or a cation?
Arrange the following in increasing order of property indicated
I, I+, I- (atomic size)
Select the correct answer
Atomic radii of fluorine and neon in angstrom unit are
Arrange the following as per the instruction given in the bracket.
Mg, Cl, Na, S, Si (increasing order of atomic size)
Arrange the following as per the instruction given in the bracket
Na, K, Li (Increasing atomic size)
On moving from left to right in a periodic table, the size of the atom _______.
_______ is the distance between the nucleus of the atom and its outermost shell.
While going from top to bottom in a group the atomic radius _______.
Moving from left to right, the size of the atom decreases.
Write information about the given atomic numbers in the table. 10, 20, 7.
| Atomic Number | Electronic configuration | Group | Period | Element |
| 10 | ||||
| 20 | ||||
| 7 |
When an atom of iodine becomes an iodine ion (I–) the radius will ______
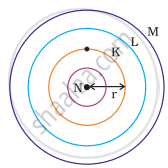
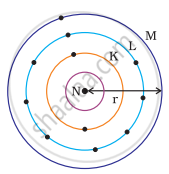
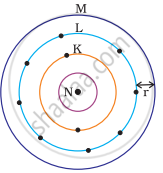
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
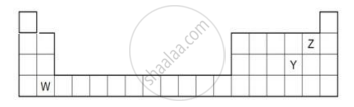
The diagram below shows part of the periodic table.
- Which elements would react together to form covalent compounds?
- Between the two elements W and Z, which will have a bigger atomic radius? Why?
Arrange the following in order of increasing radii:
Mg2+, Mg, Mg+
Explain your choice.
Arrange the following in order of increasing radii:
N, O, P
Explain your choice.
This question refers to the elements of the Periodic Table with atomic numbers from 3 to 18. Some of the elements are shown by letters, but the letters are not the usual symbols of the elements.
| 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| A | B | C | D | E | F | G | H |
| 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
| I | J | K | L | M | N | O | P |
Which of these have least atomic size in period 3?