Advertisements
Advertisements
Question
Assertion (A): At constant temperature, pV vs V plot for real gases is not a straight line.
Reason (R): At high pressure all gases have \[\ce{Z}\] > 1 but at intermediate pressure most gases have \[\ce{Z}\] < 1.
Options
Both A and R are true and R is the correct explanation of A.
Both A and R are true but R is not the correct explanation of A.
A is true but R is false.
A is false but R is true.
Solution
Both A and R are true but R is not the correct explanation of A.
Explanation:
At constant temperature, the pV vs V plot for real gases is not a straight line because there is intermolecular attraction present in real gases which is absent in ideal gases; hence ideal gases form a straight line in the pV vs V plot at constant temperature.
APPEARS IN
RELATED QUESTIONS
Maximum deviation from ideal gas is expected from
Which of the following diagrams correctly describes the behaviour of a fixed mass of an ideal gas? (T is measured in K)
In what way real gases differ from ideal gases.
Explain whether a gas approaches ideal behavior or deviates from ideal behaviour if the temperature is raised while keeping the volume constant.
Which of the following gases would you expect to deviate from ideal behavior under conditions of low-temperature F2, Cl2, or Br2? Explain.
A plot of volume (V) versus temperature (T) for a gas at constant pressure is a straight line passing through the origin. The plots at different values of pressure are shown in Figure. Which of the following order of pressure is correct for this gas?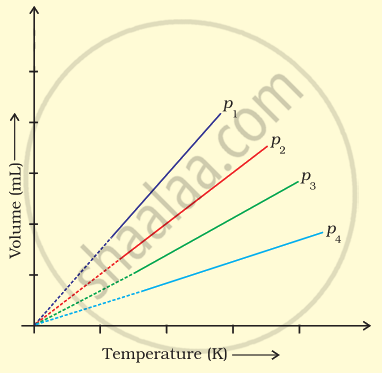
If 1 gram of each of the following gases are taken at STP, which of the gases will occupy (a) greatest volume and (b) smallest volume?
\[\ce{CO, H2O, CH4 , NO}\]
Compressibility factor, Z, of a gas is given as Z = `(pV)/(nRT)`. What is the value of Z for an ideal gas?
In van der Waal's equation for the real gas, the expression for the net force of attraction amongst the gas molecules is given by:
Choose the correct option for the total pressure (in atm.) in a mixture of 4g \[\ce{O2}\] and 2g \[\ce{H2}\] confined in a total volume of one litre at 0°C is ______.
[Given R = 0.082 L atm mol−1K−1, T = 273 K]
