Advertisements
Advertisements
Question
Maximum deviation from ideal gas is expected from
Options
\[\ce{CH4_{(g)}}\]
\[\ce{NH3_{(g)}}\]
\[\ce{H2_{(g)}}\]
\[\ce{N2_{(g)}}\]
Solution
\[\ce{NH3_{(g)}}\]
APPEARS IN
RELATED QUESTIONS
Calculate the volume occupied by 8.8 g of CO2 at 31.1°C and 1 bar pressure. R = 0.083 bar L K–1 mol–1.
Compressibility factor for CO2 at 400 K and 71.0 bar is 0.8697. The molar volume of CO2 under these conditions is
Which of the following diagrams correctly describes the behaviour of a fixed mass of an ideal gas? (T is measured in K)
25 g of each of the following gases are taken at 27°C and 600 mm Hg pressure. Which of these will have the least volume?
In what way real gases differ from ideal gases.
Write the Van der Waals equation for a real gas. Explain the correction term for pressure and volume.
A plot of volume (V) versus temperature (T) for a gas at constant pressure is a straight line passing through the origin. The plots at different values of pressure are shown in Figure. Which of the following order of pressure is correct for this gas?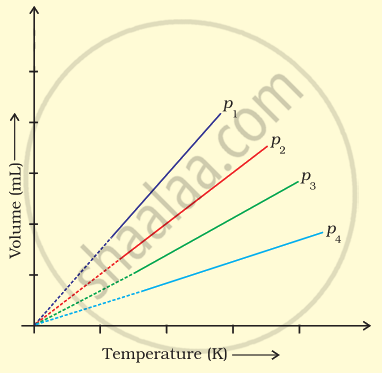
If 1 gram of each of the following gases are taken at STP, which of the gases will occupy (a) greatest volume and (b) smallest volume?
\[\ce{CO, H2O, CH4 , NO}\]
Match the following graphs of ideal gas with their co-ordinates:
| Graphical representation | x and y co-ordinates |
(i)  |
(a) pV vs. V |
(ii) 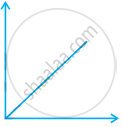 |
(b) p vs. V |
(iii) 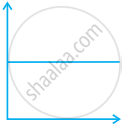 |
(c) p vs. `1/V` |
Choose the correct option for the total pressure (in atm.) in a mixture of 4g \[\ce{O2}\] and 2g \[\ce{H2}\] confined in a total volume of one litre at 0°C is ______.
[Given R = 0.082 L atm mol−1K−1, T = 273 K]
