Advertisements
Advertisements
Question
Balance the chemical equation.
\[\ce{HNO3 +Ca(OH)2 -> Ca(NO3)2 + H2O}\]
Solution
\[\ce{2HNO3 +Ca(OH)2 -> Ca(NO3)2 + 2H2O}\]
APPEARS IN
RELATED QUESTIONS
Balance the given equation:
BaCI2 + H2SO4  BaSO4 + HCI
BaSO4 + HCI
When hydrogen is passed over copper oxide, copper and steam are formed. Write a balanced equation for this reaction and state which of the chemicals are non-metals.
What do you understand by exothermic reactions?
The metal M reacts vigorously with water to form a solution S and a gas G. The solution S turns red litmus to blue whereas gas G, which is lighter than air, burns with a pop sound. Metal M has a low melting point and it is used as a coolant in nuclear reactors.
(a) What is metal M?
(b) What is solution S? Is it acidic or alkaline?
(c) What is gas G?
(d) Write a balanced chemical equation for the reaction which takes place when metal M reacts with water.
(e) Is this reaction exothermic or endothermic?
Balance the following chemical equation:
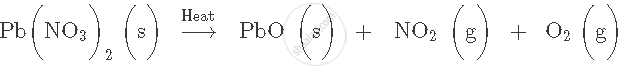
Balance the following equation. Also name the product formed.
K2O + H2O → KOH
Balance the following equation:
Zn + KOH → K2ZnO2 + H2
Sodium chloride reacts with silver nitrate to produce silver chloride and sodium nitrate
Write the equation.
Check whether it is balanced, if not balance it.
Find the weights of reactants and products.
State the law which this equation satisfies.
Write the balanced chemical equation of the following reaction.
Potassium permanganate + hydrochloric acid → potassium chloride + manganese chloride + chlorine + water
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate the volume of chlorine gas formed at S.T.P.
Considering the relationship in the first pair, complete the second paie :
AgCI : White :: CuI2 : ___________
Write word equation for the following molecular equation:
Na2SO3 + 2HCl [dil.] → 2NaCl + H2O + SO2 [g]
Representation of the results of a chemical change – is a chemical equation.
For the equation: FeCl3 + 3NH4OH 3NH4Cl + Fe(OH)3 ↓
Answer the following:
What is the indications of the arrow between the reactants and the products and of the arrow pointing downwards at the end.
Give word equation for the following chemical reaction and give the names of the product formed.
\[\ce{CuO + H2SO4 -> CuSO4 + H2O}\]
Name the following:
Two non-metal which react explosively when brought in close contact.
Balance the following simple equation:
Zn + NaOH → Na2ZnO2 + H2
Write a balanced equation for the following word equation:
Aluminium + Oxygen → Aluminium oxide
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
2H2O + 2Cl2 → 2HCl + O2
What is called thermolysis reactions?
Dil. HCl is added to Zn granules. How will you prove that chemical change has taken place here? Support your response with two arguments.
