Advertisements
Advertisements
Question
Considering the relationship in the first pair, complete the second paie :
AgCI : White :: CuI2 : ___________
Solution
AgCI : White :: CuI2 : Brown
APPEARS IN
RELATED QUESTIONS
Write the balanced chemical equation for the following reaction.
\[\ce{Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water}\]
What is wrong with the following chemical equation?
Mg + O → MgO
Correct and balance it.
How will you indicate a solution made in water in a chemical equation?
How will you indicate Exothermic reaction in a chemical equation?
Balance the gievn equation:
NaOH + H2SO4  Na2SO4 + H2O
Na2SO4 + H2O
Balance the given equation:
BaCI2 + H2SO4  BaSO4 + HCI
BaSO4 + HCI
When hydrogen is passed over copper oxide, copper and steam are formed. Write a balanced equation for this reaction and state which of the chemicals are reactants.
Convey the following information in the form of a balanced chemical equation:
"An aqueous solution of ferrous sulphate reacts with an aqueous solution of sodium hydroxide to form a precipitate of ferrous hydroxide and sodium sulphate remains in solution."
Write the balanced equations for the following reaction, and add the state symbols:
Magnesium carbonate reacts with hydrochloric acid to produce magnesium chloride, carbon dioxide and water.
Carbon monoxide reacts with hydrogen under certain conditions to form methanol (CH3OH). Write a balanced chemical equation for this reaction indicating the physical states of reactants and product as well as the conditions under which this reaction takes place.
Give one example of a chemical reaction characterised by formation of a precipitate.
When the solution of substance X is added to a solution of potassium iodide, then a yellow solid separates out from the solution.
(a) What do you think substance X is likely to be?
(b) Name the substance which the yellow solid consists of.
(c) Which characteristic of chemical reaction is illustrated by this example?
(d) Write a balanced chemical equation for the reaction which takes place. Mention the physical states of all the reactant and products involved in the chemical equation.
The metal M reacts vigorously with water to form a solution S and a gas G. The solution S turns red litmus to blue whereas gas G, which is lighter than air, burns with a pop sound. Metal M has a low melting point and it is used as a coolant in nuclear reactors.
(a) What is metal M?
(b) What is solution S? Is it acidic or alkaline?
(c) What is gas G?
(d) Write a balanced chemical equation for the reaction which takes place when metal M reacts with water.
(e) Is this reaction exothermic or endothermic?
When a mixture of gases X and Y is compressed to 300 atm pressure and then passed over a catalyst consisting of a mixture of zinc oxide and chromium oxide (heated to a temperature of 300°C), then an organic compounds Z having the molecular formula CH4O is formed. X is a highly poisonous gas which is formed in appreciable amounts when a fuel burns in a limited supply of air; Y is a gas which can be made by the action of a dilute acid on an active metal; and Z is a liquid organic compound which can react with sodium metal to produce hydrogen gas.
(a) What are X, Y and Z?
(b) Write a balanced chemical equation of the reaction which takes place when X and Y combine to form Z. Indicate the conditions under which the reaction occurs.
Balance the following chemical equation :
Na2CO3 + HCl → NaCI + H2O + CO2
Balance the following equation. Also name the product formed.
PbCO3 → PbO +CO2
In electrolysis of water, why is the volume of gas collected over one electrode double that of gas collected over the other electrode?
Show the formation of Na2O by the transfer of electrons.
Write down what you understood from the following chemical reaction.
AgNO3(aq) + NaCI(aq) → AgCI ↓ + NaNO3(aq)
Balance the following equation:
MnO2 + HCl → MnCl2 + H2O + Cl2
Balance the following equation:
C + H2SO4 → CO2 + H2O + SO2
What information do the following chemical equations convey? Mg + 2HCl → MgCl2+ H2
Choose the correct answer from the options given below.
The formula of sodium carbonate is Na2CO3 and that of calcium hydrogen carbonate is
Write the balanced chemical equation of the following reaction. potassium bicarbonate + sulphuric acid → potassium sulphate + carbon dioxide + water
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate moles of salt formed.
When a potassium iodide solution is added to a solution of lead (II) nitrate in a test tube, a precipitate is formed.
Write the balanced chemical equation for this reaction.
Write word equation for the following molecular equation:
AgN03 + HCI → HNO3 + AgCl ↓
Word equation:
State whether AgCl is soluble or insoluble in HN03.
Write word equation for the following molecular equation:
CaO + H20 → Ca (OH)2 + Δ
Word equation:
State why [+ Δ ] is not written in the reaction, if ZnO reacts with H2O.
In certain reaction a change of state is observed i.e. solid to liquid, liquid to gas etc. – State the change of state of the products – to give the respective reactant.
\[\ce{2H2O ->[electric][current] 2H2 + O2}\]
Representation of the results of a chemical change – is a chemical equation.
For the equation: FeCl3 + 3NH4OH 3NH4Cl + Fe(OH)3 ↓
Answer the following:
Is the equation given above a balanced equation. Give reasons.
Give an example of a chemical equation in which two reactants form:
four product
Balance the following simple equation:
Al + N2 → AlN
Balance the following simple equation:
KBr + Cl2 → KCl + Br2
Balance the following simple equation:
NaOH + Cl2 → NaCl + NaClO + H2O
Balance the following simple equation:
CaO + HCl → CaCl2 + H2O
Writing a chemical reaction in brief by using chemical formulae is called as _______.
What is a chemical equation?
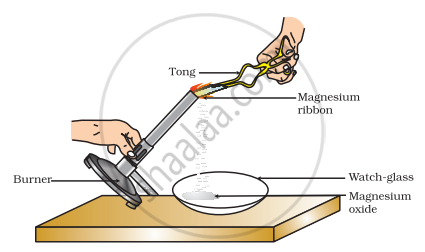
Which of the following is the correct observation of the reaction shown in the above set up?
To balance the following chemical equation the value of x and y should respectively be:
\[\ce{2NaOH + xAl_2O_3->yNaAlO_2 + H_2O}\]
