Advertisements
Advertisements
Question
Choose the correct option.
Hyperconjugation involves overlap of ______ orbitals.
Options
σ - σ
σ -p
p - p
π - π
Solution
Hyperconjugation involves overlap of σ - p orbitals.
APPEARS IN
RELATED QUESTIONS
Draw a resonance structure of the following:
Phenol
Draw a resonance structure of the following:
Buta-1,3-diene
Choose the correct option.
Which of the following statements are true with respect to electronic displacement in a covalent bond?
a. Inductive effect operates through π bond
b. Resonance effect operates through σ bond
c. Inductive effect operates through σ bond
d. Resonance effect operates through π bond
The correct IUPAC name of the compound 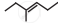 is ______.
is ______.
Choose the correct option.
The geometry of a carbocation is ______.
Which of the following is NOT an electrophile?
Which of the following is the strongest nucleophile?
Which of the following statements is INCORRECT about hyperconjugation?
Which among the following is a set of nucleophiles?
Which of the following is TRUE for homolytic fission?
Identify the reagent used in the following reaction:
\[\ce{CH3 - CH2 - Br ->[?] CH3 - CH2 - OH}\]
The best reagent for the following conversion is:
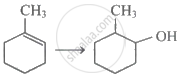
Which of the following is the most unstable carbocation?
IUPAC name of ![]() is ______.
is ______.
How many pi bonds and sigma bonds are present in following molecule?
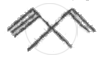
Which of the following statements is not correct?
Which element among the following does form pπ - pπ multiple bonds?
Arrange the following free radicals in order of decreasing stability.
- Methyl
- Vinyl
- Allyl
- Benzyl
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen in each.
\[\ce{CH3 - CH2 - \overset{\oplus}{C}H -CH2 - CH2 }\]
Identify the α-carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
CH2 = CH - CH2 - CH3
Identify the α - carbons in the following species and give the total number of α-hydrogen in each.
\[\ce{CH3 - CH2 - \overset{\oplus}{C}H - CH2 - CH3}\]
