Advertisements
Advertisements
Question
Compressibility factor, Z, of a gas is given as Z = `(pV)/(nRT)`. For real gas what will be the effect on value of Z above Boyle’s temperature?
Solution
For real gas, the value of Z > 1 above Boyle's temperature .
APPEARS IN
RELATED QUESTIONS
Compressibility factor for CO2 at 400 K and 71.0 bar is 0.8697. The molar volume of CO2 under these conditions is
Maximum deviation from ideal gas is expected from
Which of the following diagrams correctly describes the behaviour of a fixed mass of an ideal gas? (T is measured in K)
25 g of each of the following gases are taken at 27°C and 600 mm Hg pressure. Which of these will have the least volume?
In what way real gases differ from ideal gases.
Can a Van der Waals gas with a = 0 be liquefied? explain.
Explain whether a gas approaches ideal behavior or deviates from ideal behaviour if it is compressed to a smaller volume at a constant temperature.
Which of the following gases would you expect to deviate from ideal behavior under conditions of low-temperature F2, Cl2, or Br2? Explain.
Under which of the following two conditions applied together, a gas deviates most from the ideal behaviour?
(i) Low pressure
(ii) High pressure
(iii) Low temperature
(iv) High temperature
Match the following graphs of ideal gas with their co-ordinates:
| Graphical representation | x and y co-ordinates |
(i)  |
(a) pV vs. V |
(ii) 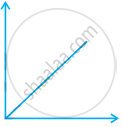 |
(b) p vs. V |
(iii) 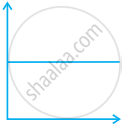 |
(c) p vs. `1/V` |
