Advertisements
Advertisements
Question
Derive the relationship between Relative molecular mass and Vapour density.
Solution
(i) The Relative Molecular Mass of a gas or vapour is the ratio between the mass of one molecule of the gas or vapour to the mass of one atom of Hydrogen.
(ii) Vapour density is the ratio of the mass of a certain volume of a gas or vapour, to the mass of an equal volume of hydrogen, measured under the same conditions of temperature and pressure.
Vapour Density (V.D.) = `"Mass of a given volume of gas or vapour at S.T.P"/"Mass of the same volume of hydrogen"`.
(iii) According to Avogadro’s law, equal volumes of all gases contain an equal number of molecules. Thus, let the number of molecules in one volume = n, then
(iv) V.D at STP = `" Mass of molecules of a gas or vapour at S.T.P"/"Mass of 'n' ‘molecules of hydrogen"`
Cancelling ‘n’ which is common, you get
V.D = `"Mass of 1 molecule of a gas or vapour at S.T.P."/"Mass of 1 molecule of hydrogen"`.
(v) Since hydrogen is diatomic
V.D. = `"Mass of 1 molecule of a gas or vapour at S.T.P."/"Mass of 2 atoms of hydrogen"`
(vi) By comparing the definition of relative molecular mass and vapour density we can write as follows.
V.D. = `"Mass of 1 molecule of a gas or vapour at S.T.P."/(2 × "Mass of 1 atom of hydrogen")`
Relative molecular mass (hydrogen scale) = `"Mass of 1 molecule of a gas or vapour at STP."/"Mass of 1 atom of hydrogen"`
(vii) By substituting the relative molecular mass value in vapour density definition, we get
Vapour density (V.D.) = Relative molecular mass/2
⇒ 2 × vapour density = Relative molecular mass of a gas.
APPEARS IN
RELATED QUESTIONS
Write down the name of a compound represented by the following formula:
CaCl2
Give the name of the element present in the following compound:
Quick lime
Give the name of the element present in the following compound:
Baking powder
Give the name of the element present in the following compound:
Potassium sulphate
State the term.
The number of atoms present in a molecule of an element _________
Define: Atomic number
What is meant by atomicity ? Explain with two examples.
In what form does oxygen gas occur in nature ?
What do the following denote ?
- N
- 2N
- N2
- 2N2
What is the difference between the molecule of an element and the molecule of a compound ? Give one
example of each.
Work out the formula for magnesium hydrogencarbonate.
The cation of an element has :
Molecular compounds are usually formed by the combination between :
Give one example of a triatomic molecule
State the main points of the contradiction of Dalton’s atomic theory by the Modern Atomic Theory.
Give a reason why –
Helium is considered monoatomic, but hydrogen a diatomic molecule.
Give a reason why –
Ozone is considered triatomic, but phosphorus a tetratomic molecule.
State the atomicity of the following:
Chlorine
State the atomicity of the following:
Argon
Which of the following is a short and scientific way of representing one molecule of an element or compound?
Classify the following molecules as the molecules of element or compound
| 1. |  |
|
| 2. |  |
|
| 3. | 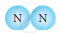 |
|
| 4. | 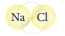 |
Define: Atomicity.
Give any two examples for heterodiatomic molecules.
