Advertisements
Advertisements
Question
Describe Freundlich adsorption isotherm.
Solution 1

Freundlich adsorption isotherm:
Freundlich adsorption isotherm gives an empirical relationship between the quantity of gas adsorbed by the unit mass of solid adsorbent and pressure at a specific temperature.
From the given plot it is clear that at pressure PS, x/m reaches the maximum valve. Ps is called the saturation pressure. Three cases arise from the graph now.
Case I- At low pressure:
The plot is straight and sloping, indicating that the pressure in directly proportional to x/m i.e., `x/m alpha P`
`x/m = kP` (k is constant)
Case II- At high pressure:
When pressure exceeds the saturated pressure, x/m becomes independent of P values
`x/m alpha P^@`
`x/m = kP^@`
Case III- At intermediate pressure:
At intermediate pressure, x/m depends on P raised to the powers between 0 and 1. This relationship is known as the Freundlich adsorption isotherm.
`x/m alpha P^(1/n)`
`x/m = kP^(1/n)` n > 1
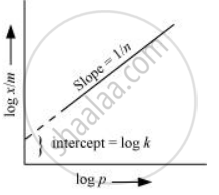
Now taking log: `log x/m =log k + 1/n log P`
On plotting the graph between log (x/m) and log P, a straight line is obtained with the slope equal to 1/n and the intercept equal to log k
Solution 2
Freundlich Adsorption isotherm:
Freundlich, in 1909, gave an empirical relationship between the quantity, of gas adsorbed by unit mass of solid adsorbent and pressure at a particular temperature. The relationship can be expressed by the following equation:
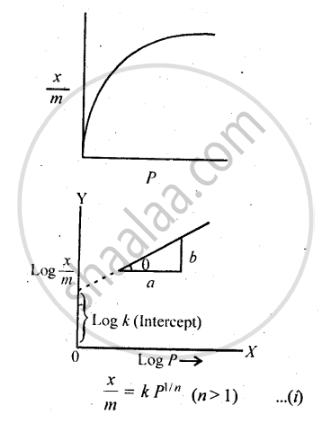
where x is the mass of the gas adsorbed by mass ‘m’ of the adsorbent at pressure P, k and n are constants which depend on the nature of the adsorbent and the gas at a particular temperature. The relationship is generally represented in the form of a curve where mass of the gas adsorbed per gram by the adsorbent is plotted against pressure. These curves indicate that at a fixed pressure, there is a decrease in physical adsorption with increase in temperature. These curves always seem to approach saturation at high pressure.
Taking log of equation (i), we get
`log x/m =log k + 1/n log P`
APPEARS IN
RELATED QUESTIONS
What is an adsorption isotherm?
What do you understand by activation of adsorbent? How is it achieved?
In reference to Freundlich adsorption isotherm, write the expression for adsorption of gases on solids in the form of an equation.
Explain graphically Freundlich adsorption isotherm.
The slope and intercept for plot of `log_10 "x"/"m"` against log10P for Freundlich equation are ____________ and ____________ respectively.
Freundlich's equation for adsorption of gas on solid is represented as ____________.
In Freundlich's adsorption isotherm, when log `("x"/"m")` is plotted against log P, slope of the graph is ____________.
Freundlich adsorption isotherm is given by the expression `x/m = kp^(1/n)` which of the following conclusions can be drawn from this expression.
(i) When `1/n` = 0, the adsorption is independent of pressure.
(ii) When `1/n` = 0, the adsorption is directly proportional to pressure.
(iii) When n = 0, `x/m` vs p graph is a line parallel to x-axis.
(iv) When n = 0, plot of `x/m` vs p is a curve.
Adsorption of gases on the solid surface is generally exothermic because.
In the freundilch adsorption isotherm, the value of x/m is 0.4 under a pressure of 0.2 at m calculate the value of the interact
Which of the following statement is INCORRECT regarding the adsorption of a gas on the surface of the solid?
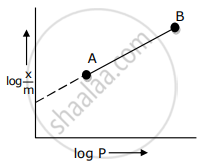
In Freundlich adsorption isotherm, slope of AB line is:
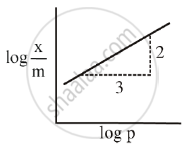
The mass of gas adsorbed, x per unit mass of adsorbed, m was measured at various x pressures p. A graph between `log "x"/"m"` and log p gives a straight line with slope equal to 2 and the intercept equal to 0.4771. The value of `"x"/"m"` at a pressure of 4 atm is ______.
[Given log 3 = 0.4771]
Adsorption of a gas follows Freundlich adsorption isotherm. x is the mass of the gas adsorbed on mass m of the adsorbent. The x plot of log `x/m` versus log p is shown in the given graph. `x/m` is proportional to ______.
For Freundlich adsorption isotherm, a plot of log (x/m) (Y-axis) and log p (x-axis) gives a straight line. The intercept and slope for the line is 0.4771 and 2, respectively. The mass of gas, adsorbed per gram of adsorbent if the initial pressure is 0.04 atm, is ______ × 10-4 g. (log 3 = 0.4771)
