Advertisements
Advertisements
Question
Does an ideal gas exist in practice?
Solution
No, ideal gases do not exist in practice. The concept of an ideal gas is a theoretical model used to simplify the study of gases. However, real gases can behave very similarly to ideal gases under certain conditions.
APPEARS IN
RELATED QUESTIONS
Find the kinetic energy of 3 litre of gas at S.T.P given standard pressure is 1.013 × 105 N/m2.
Determine the pressure of nitrogen at 0°C if the density of nitrogen at N.T.P. is 1.25 kg/m3 and R.M.S. speed of the molecules at N.T.P. is 489 m/s.
A sample of an ideal gas is at equilibrium. Which of the following quantity is zero?
Two identically sized rooms A and B are connected by an open door. If the room A is air-conditioned such that its temperature is 4°C lesser than room B, which room has more air in it?
If the internal energy of an ideal gas U and volume V are doubled then the pressure ____________.
The ratio γ = `"C"_"p"/"C"_"v"` for a gas mixture consisting of 8 g of helium and 16 g of oxygen is ____________.
Which of the following shows the correct relationship between the pressure and density of an ideal gas at constant temperature?
The following graph represents the pressure versus number density for an ideal gas at two different temperatures T1 and T2. The graph implies
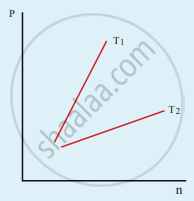
What is the microscopic origin of pressure?
A gas is at temperature 80°C and pressure 5 × 10−10 Nm−2. What is the number of molecules per m3 if Boltzmann’s constant is 1.38 × 10−23 J K−1
Estimate the total number of air molecules in a room of a capacity of 25 m3 at a temperature of 27°C.
A perfect gas of 'N' molecules, each of mass 'm', moving with velocities 'C1', 'C2', ...... .'CN' is enclosed in a cubical vessel of volume 'V'. The pressure exerted by the gas on the walls of the vessel is ______. ('p' = density of gas)
The average force applied on the walls of a closed container depends as 'Tx', where 'T' is the temperature of an ideal gas. The value of 'x' is ______.
What is an ideal gas?
The kinetic energy per molecule of a gas at temperature T is ______.
The velocities of five molecules are 2 m/s, 3 m/s, 4 m/s, 5 m/s and 6 m/s. Find the root mean square velocity of molecules.
