Advertisements
Advertisements
Question
Draw the structures of BCl3.NH3 and AlCl3 (dimer).
Solution
In BCl3, the central B atom has six electrons in the valence shell. It is, therefore, an electron-deficient molecule and needs two more electrons to ‘ complete its octet. In other words, BCl3 acts as a Lewis acid. NH3, on the other hand, has a lone pair of electrons which it can donate easily. Therefore, NH3 acts as a Lewis base. The Lewis acid (BC13) and the Lewis base (NH3) combine together to form an adduct as shown below:
In AlC13, Al has six electrons in the valence shell. Therefore, it is an electron-deficient molecule and needs two more electrons to complete its octet. Chlorine, on the other hand, has three lone pairs of electrons. Therefore, to complete its octet, the central Al atom of one molecule accepts a lone pair of electrons from Cl atom of the other molecule forming a dimeric structure as shown below.
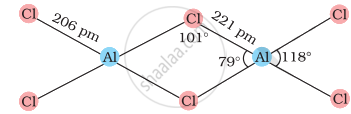
AlCl3 (dimer)
APPEARS IN
RELATED QUESTIONS
Is boric acid a protic acid? Explain.
Explain what happens when boric acid is heated.
Explain the Structure of Boric Acid.
A certain salt X, gives the following results.
- Its aqueous solution is alkaline to litmus.
- It swells up to a glassy material Y on strong heating.
- When conc. H2SO4 is added to a hot solution of X, white crystal of an acid Z separates out.
Write equations for all the above reactions and identify X, Y and Z.
Write a balanced equation for
Boric acid is an acid because its molecule ______.
When aqueous solution of borax is acidified with hydrochloric acid, a white crystalline solid is formed which is soapy to touch. Is this solid acidic or basic in nature? Explain.
Which of the following compounds are formed when boron trichloride is treated with water?
On the addition of mineral acid to an aqueous solution of borax, the compound formed is ______.
Boron reacts with nitric acid to form ______.
