Advertisements
Advertisements
Question
Explain Boyle's Law on the basis of the kinetic theory of matter.
Solution
Boyle's law on the basis of the kinetic theory of matter:
- According to the kinetic theory of matter, the number of particles present in a given mass and the average kinetic energy is constant.
- If the volume of the given mass of a gas is reduced to half of its original volume, then the same number of particles will have half the space to move.
- As a result, the number of molecules striking the unit area of the walls of the container at a given time will double and the pressure will also double.
- Alternatively, if the volume of a given mass of a gas is doubled at a constant temperature, the same number of molecules will have double the space to move.
- Thus, the number of molecules striking the unit area of the walls of a container at a given time will become one-half of the original value.
- Thus, the pressure will also get reduced to half of the original pressure. Hence, it is seen that if the pressure increases, the volume of a gas decreases at a constant temperature, which is Boyle's law.
APPEARS IN
RELATED QUESTIONS
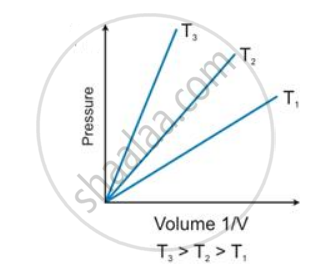
State the law which is represented by the following graph:
A steel cylinder of internal volume 20 litres is filled with hydrogen at 29 atmospheric pressure. If hydrogen is used to fill a balloon at 1.25 atmospheric pressure at the same temperature, what volume will the gas occupy?
A certain mass of a gas occupies 2 litres at 27°C and 100 Pa. Find the temperature when volume and pressure become half of their initial values.
Calculate the volume occupied by 2 g of hydrogen at 27°C and 4-atmosphere pressure if at STP it occupies 22.4 litres.
Calculate the volume of a gas ‘A’ at s.t.p., if at 37°C and 775 mm of mercury pressure, it occupies a volume of `9 1/2` litres.
Calculate the following:
Calculate the temperature at which a gas ‘A’ at 20°C having a volume, of 500 cc. will occupy a volume of 250 cc.
Calculate the following:
A gas ‘X’ is collected over water at 17°C and 750 mm. pressure. If the volume of the gas collected is 50 cc., calculate the volume of the dry gas at s.t.p. [at 17°C the vapour pressure is 14 mm.]
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 380 lits. originally occupying 800 cm3 at 76 cm. pressure.
According to Boyle’s law, the shape of the graph between pressure and reciprocal of volume is _______.
State-the law of volume
