Advertisements
Advertisements
Question
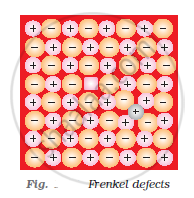
Explain the following terms with suitable examples: Frenkel defect
Solution
Frenkel defect: Ionic solids containing large differences in the sizes of ions show this type of defect. When the smaller ion (usually cation) is dislocated from its normal site to an interstitial site, Frenkel defect is created. It creates a vacancy defect as well as an interstitial defect. Frenkel defect is also known as dislocation defect. Ionic solids such as AgCl, AgBr, AgI, and ZnS show this type of defect.
APPEARS IN
RELATED QUESTIONS
Give reasons : In stoichiometric defects, NaCl exhibits Schottky defect and not Frenkel defect.
Examine the given defective crystal:

Answer the following questions :
(i) What type of stoichiometric defect is shown by the crystal?
(ii) How is the density of the crystal affected by this defect?
(iii) What type of ionic substances show such defect?
non-stoichiometric defects?
Out of NaCl and AgCl, which one shows Frenkel defect and why?
Answer the following question.
What type of stoichiometric defect is shown by ZnS and why?
Ionic solids containing large differences in sizes of ions show ____________.
Defects in solids can be studied using
Point defect is also known as ____________.
Type of stoichiometric defect shown by AgBr is ____________.
The following diagram shows:
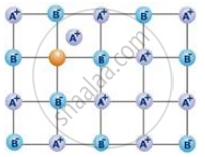
Assertion: No compound has both Schottky and Frenkel defects.
Reason: Both defects change the density of the solid.
Alkali halids do not show Frenkel defect because ____________.
Which of the following crystals does not exhibit Frenkel defect?
Silver halides generally show:
Which of the following statements are not true?
(i) Vacancy defect results in a decrease in the density of the substance.
(ii) Interstitial defects results in an increase in the density of the substance.
(iii) Impurity defect has no effect on the density of the substance.
(iv) Frankel defect results in an increase in the density of the substance.
Which of the following defects decrease the density?
(i) Interstitial defect
(ii) Vacancy defect
(iii) Frankel defect
(iv) Schottky defect
Why is FeO (s) not formed in stoichiometric composition?
If NaCl is doped with 10–4 mole percent of SrCl2 the concentration of cation vacancies will be
Schottky defect is noticed in ______
The incorrect statement about the imperfections in solids is ______.
