Advertisements
Advertisements
Question
Explain why does conductivity of germanium crystals increase on doping with galium.
Solution
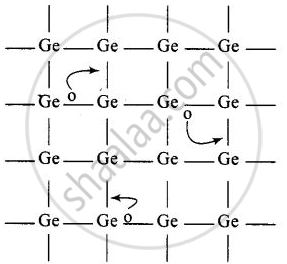
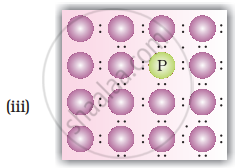
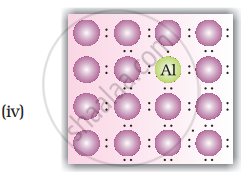
- When Ge is doped with group 13 elements, for example, gallium, the structure of crystal lattice does not change.
- 3 valence electrons of gallium are used up in the normal covalent bond.
- For one dopant atom, one hole is created because the place where fourth electron is missing is called vacancy or hole and is responsible for conduction of germanium doped with gallium.
Electron from neighbouring atom comes and fills the hole, thereby creating a hole in its original position. Under the influence of electric field electrons move towards positively charged plates through these and conduct electricity. The holes appear to move towards negatively charged plates.
APPEARS IN
RELATED QUESTIONS
p - type semi-conductors are made by mixing silicon with impurities of-
- germanium
- boron
- arsenic
- antimony
A pure semiconductor is ________.
(a) an extrinsic semiconductor
(b) an intrinsic semiconductor
(c) p-type semiconductor
(d) n-type semiconductor
Distinguish between p-type and n-type semiconductors.
Group 14 element is converted to p-type semiconductor by doping it with:
In n-type semiconductor, current is carried by ____________.
p-type semiconductors are formed when Si or Ge are doped with ____________.
Which of the following is true about the charge acquired by p-type semiconductors?
Under the influence of electric field, which of the following statements is true about the movement of electrons and holes in a p-type semiconductor?
(i) Electron will move towards the positvely charged plate through electron holes.
(ii) Holes will appear to be moving towards the negatively charged plate.
(iii) Both electrons and holes appear to move towards the positively charged plate.
(iv) Movement of electrons is not related to the movement of holes.
A perfect crystal of silicon (Figure) is doped with some elements as given in the options. Which of these options show n-type semiconductors?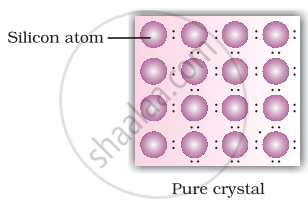
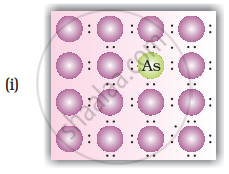
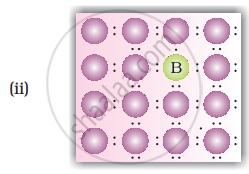
How does the doping increase the conductivity of semiconductors?
