Advertisements
Advertisements
Question
Explain Wolf-Kishner reduction reaction.
Solution
The carbonyl group of aldehydes and ketones is reduced to the methylene group \[\ce{[- CH2 -]}\] on treatment with hydrazine followed by heating with sodium or potassium hydroxide in a high-boiling solvent like ethylene glycol, known as the Wolf-Kishner reduction.
| \[\begin{array}{cc} \ce{O}\phantom{...................................}\\ ||\phantom{.....................}\backslash\phantom{.............}\\ \ce{C + \underset{(Hydrazine)}{NH2 - NH2} ->[-H2O] \underset{(Hydrazone)}{C = N - NH2}}\\ /\phantom{..}\backslash\phantom{....................}/\phantom{..............}\\ \end{array}\] |
| \[\begin{array}{cc} \backslash\phantom{.............................}\backslash\phantom{..............}\\ \ce{\underset{(Hydrazone)}{C = N - NH2} ->[KOH, \Delta][Ethylene glycol] \underset{(Methyl group)}{CH2} + N2}\\ /\phantom{.............................}/\phantom{..............}\\ \end{array}\] |
Here the oxygen of the carbonyl group is replaced by two hydrogen atoms.
APPEARS IN
RELATED QUESTIONS
Write reaction showing aldol condensation of cyclohexanone.
The following compounds will give positive Fehling’s test.
Write the name of the product when ketones react with 1,2-diol in presence of dry HCl.
Which of the following is Schiff's reagent?
Identify A in the following reaction.
\[\ce{A + CH3MgBr ->[Ether] complex ->[H3O+] (CH3)3C - OH}\]
ln which of the following reactions, both oxidised and reduced forms of the same compound are obtained?
Compounds of general formula, are called ____________.
\[\begin{array}{cc} \phantom{...}\ce{R}\phantom{....}\ce{OR''}\phantom{}\\ \phantom{}\backslash\phantom{..}/\\ \ce{C}\\ \phantom{}/\phantom{..}\backslash\\ \phantom{...}\ce{R'}\phantom{...}\ce{OR''}\phantom{} \end{array}\]
Which of the following carbonyl compounds does NOT undergo aldol condensation?
Which of the following reagents is used for the following conversion?
\[\ce{CH3 - CH = CH - CHO -> CH3 - CH = CH - CH2OH}\]
\[\ce{Benzoic acid ->[i) NH3][ii) \Delta] A ->[NaOBr] B ->[NaNO2/HCl] C}\] ‘C’ is:
\[\ce{CH3Br ->[KNC] (A) ->[H3O^+] (B) ->[PCl5] (C)}\] product (C) is:
In which case, chiral carbon is not generated by reaction with HCN.
Which one of the following reaction is an example of disproportionation reaction.
What is the action of HCN on propanone?
How is the following conversion effected phenyl methanal into benzoic acid?
How will you prepare acetic anhydride from acetic acid?
How will you prepare ethyl acetate from methyl acetate?
How will you prepare acetamide from methyl cyanide?
How will you prepare lactic acid from ethanol?
How will you prepare acetophenone from acetyl chloride?
Which of the following compounds will undergo self-aldol condensation in the presence of cold dilute alkali?
A substance C4H10O yields on oxidation a compound, C4H8O which gives an oxime and a positive iodoform test. The original substance on treatment with a conc. H2SO4 gives C4H8. The structure of the compound is ______.
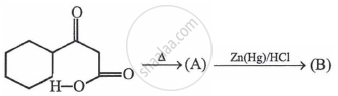
In the above reaction, product (B) is:
What is the number of different products formed when mixture of ethanal and propanal reacts with aq. NaOH after warming?
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
What is the action of hydrogen cyanide on the following?
Acetone
Identify B in the following reaction:
\[\begin{array}{cc}
\ce{O}\phantom{........}\\
||\phantom{........}\\
\ce{\underset{(Acetone)}{CH3 - C - CH3}->[CrO3] B}
\end{array}\]
What is the action of the following regent on propanal?
Sodium Bisulphite
