Advertisements
Advertisements
Question
For the first order thermal decomposition reaction, the following data were obtained:
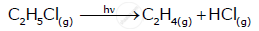
Time / sec Totalpressure / atm
0 0.30
300 0.50
Calculate the rate constant
(Given: log 2 = 0.301, log3 = 0.4771, log 4 = 0.6021)
Solution
Given :
Pi = 0.30 atm
Pt = 0.50 atm
C2H5Cl(g)→C2H4(g)+HCl(g)
Pi 0 0 (At t = 0 sec)
Pi-x x x (At t = 300 sec)
So,
Pi - x + x + x = Pt
0.3+x = 0.5
x = 0.2
Then, Pi - x = 0.3 - 0.2 = 0.1 atm
For first order reaction,
`k=2.303/tlog(P_i/(P_i-x))`
`=2.303/tlog(0.3/0.1)`
`=(2.303xxlog3)/300=(2.303xx0.4771)/300`
k = 0.0037 s-1
APPEARS IN
RELATED QUESTIONS
What is pseudo first order reaction? Give one· example of it.
Write molecularity of the following reaction:
2NO(g)+O2(g)→2NO2(g)
The conversion of molecules X to Y follows second order kinetics. If concentration of X is increased to three times how will it affect the rate of formation of Y?
Which of the following statements is not correct about order of a reaction.
The value of rate constant of a pseudo first order reaction ______.
In any unimolecular reaction:
(i) only one reacting species is involved in the rate determining step.
(ii) the order and the molecularity of slowest step are equal to one.
(iii) the molecularity of the reaction is one and order is zero.
(iv) both molecularity and order of the reaction are one.
Why does the rate of any reaction generally decreases during the course of the reaction?
Assertion: The enthalpy of reaction remains constant in the presence of a catalyst.
Reason: A catalyst participating in the reaction, forms different activated complex and lowers down the activation energy but the difference in energy of reactant and product remains the same.
The role of a catalyst is to change
On heating compound (A) gives a gas (B) which is constituent of air. The gas when treated with H2 in the presence of catalyst gives another gas (C) which is basic in nature, (A) should not be ______.
