Advertisements
Advertisements
Question
Give reasons for the following : Boiling point of ethanol is higher in comparison to methoxymethane.
Solution
Ethanol forms intermolecular hydrogen bonds due to the presence of hydrogen attached to the electronegative oxygen atom. As a result, ethanol exists as associated molecules while methoxymethane does not. Hence, the boiling point of ethanol is higher than that of methoxymethane.
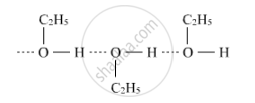
RELATED QUESTIONS
Give reasons for the following: Butan-1-ol has a higher boiling point than diethyl ether.
Give reason for the higher boiling point of ethanol in comparison to methoxymethane.
Identify the chiral molecule in the following pair :
Alcohols of low molecular weight are _____________.
Which statement is not correct about alcohol?
Assertion: Boiling points of alcohols and ethers are high.
Reason: They can form intermolecular hydrogen-bonding.
Arrange the following in the increasing order of their property indicated:
ethanol, ethanoic acid, benzoic acid (boiling point)
How are the following conversion carried out?
Methyl magnesium bromide →2-Methylpropan-2-ol.
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
How is the following conversion carried out?
Methyl magnesium bromide → 2-Methylpropan-2-ol.
