Advertisements
Advertisements
Question
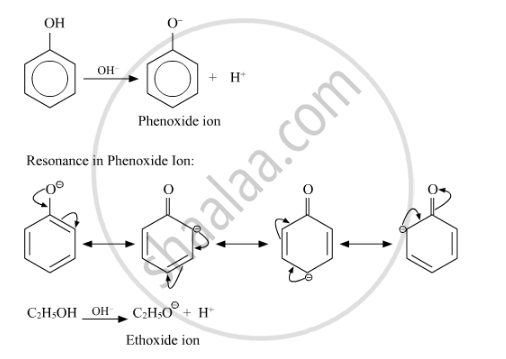
Give reasons for the following : Phenol is more acidic than ethanol.
Solution
The phenoxide ion, produced by losing of proton by phenol, is stabilised by resonance due to delocalisation of the negative charge on the benzene ring. Ethoxide ion, however, is not stabilised by resonance. On the other hand, it is further destabilised by positive inductive effect of alkyl group.
RELATED QUESTIONS
Explain why propanol has higher boiling point than that of the hydrocarbon, butane?
Give simple chemical tests to distinguish between the following pairs of compounds :
Ethanol and phenol
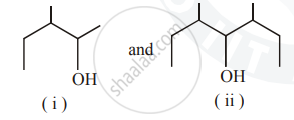
Identify the chiral molecule in the following pair :
Write the structures of A, B and C in the following reactions :
\[ C_6 H_5 {NO}_2 \to^{Sn/HCI} A \to^{{NaNO}_2 /HCI}_{273 K} B \to^{H_2 O}_∆ C\]
Alcohols have high boiling points because of ____________.
The correct order of boiling points for primary (1°), secondary (2°) and tertiary alcohol (3°) is:
A solution of phenol in chloroform when treated with aqueous NaOH gives compound P as a major product. The mass percentage of carbon in P is ______. (to the nearest integer) (Atomic mass: C = 12; H = 1; O = 16)
Assertion (A): Alcohols react both as nucleophiles and electrophiles.
Reason (R): The bond between C–O is broken when alcohols react as nucleophiles.
Select the most appropriate answer from the options given below:
How are the following conversion carried out?
Methyl magnesium bromide →2-Methylpropan-2-ol.
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
