Advertisements
Advertisements
Question
How do we obtain characteristic x-ray spectra?
Solution
Characteristic x-ray spectra:
1. X-ray spectra snow some narrow peaks at some, well-defined wavelengths when the target is hit by fast electrons. The line spectrum showing these peaks is called the characteristic x-ray spectrum.
2. When an energetic electron removes electrons from K-shell electrons than the electrons from the outer orbit jump to fill the vacancy.
3. Energy difference between the levels is given out as x-ray photons of definite wavelength.
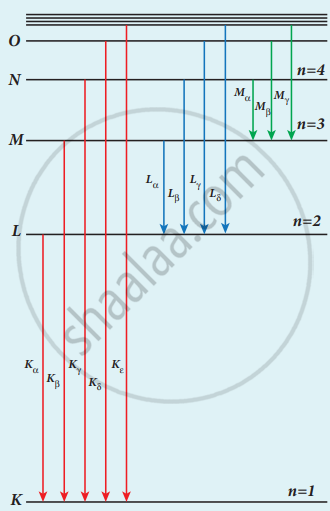
Origin of characteristic x-ray spectra
K-Series (Kα and Kβ): This line is due to electronic transitions from L, M, and N shells to K-level.
L-Series (Lα and Lβ): It arises due to electronic transitions from M, N, and O shells.
APPEARS IN
RELATED QUESTIONS
In an electron microscope, the electrons are accelerated by a voltage of 14 kV. If the voltage is changed to 224 kV, then the de Broglie wavelength associated with the electrons would
Write the expression for the de Broglie wavelength associated with a charged particle of charge q and mass m, when it is accelerated through a potential V.
A proton and an electron have the same kinetic energy. Which one has a greater de Broglie wavelength? Justify.
An electron and an alpha particle have the same kinetic energy. How are the de Broglie wavelengths associated with them related?
Explain why photoelectric effect cannot be explained on the basis of wave nature of light.
Derive an expression for de Broglie wavelength of electrons.
Describe briefly Davisson – Germer experiment which demonstrated the wave nature of electrons.
What should be the velocity of the electron so that its momentum equals that of 4000 Å wavelength photon.
Calculate the de Broglie wavelength of a proton whose kinetic energy is equal to 81.9 × 10–15 J.
(Given: mass of proton is 1836 times that of electron).
An electron is accelerated through a potential difference of 81 V. What is the de Broglie wavelength associated with it? To which part of the electromagnetic spectrum does this wavelength correspond?
