Advertisements
Advertisements
Question
Out of the three metals A, B and C of specific heat 900 J kg-1 °C-1, 380 J kg-1 °C-1 and 460 J kg-1 °C-1 respectively, which will you prefer for calorimeter? Given reason.
Solution
Heat capacity of the calorimeter should be low so out of three metals the one which have lowet specific heat capacity should be preferred.
APPEARS IN
RELATED QUESTIONS
(a) Calculate the heat capacity of a copper vessel of mass 150g if the specific heat capacity of copper is `410 J kg^-1 K^-1`
(b) How much heat energy will be required to increase the temperature of the vessel in part (a) from 25°C to 35°C?
A piece of iron of mass 2.0 kg has a heat capacity of 966 J K-1. Find heat energy needed to warm it by 15°C.
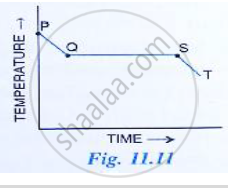
Fig 11. 11 shows the variation in temperature with time when some wax cools from the liquid phase to the solid phase.
(i) In which part of the curve, the wax is in liquid phase?
(ii) What does the part QS of the curve represent?
(iii) In which part of the curve, the wax will be the in the liquid as well as solid phase?
(iv) In which part of the curve, the wax is in solid phase?
The amount of heat energy required to convert 1 kg of ice at – 10℃ to water at 100℃ is 7,77,000 J. Calculate the specific latent heat of ice. Specific heat capacity of ice = 2100 J kg-1K-1, Specific heat capacity of water = 4200 J kg-1 K-1.
Explain, why is water used as a coolent in motor car radiators?
250 g of water at 30 °C is present in a copper vessel of mass 50 g. Calculate the mass of ice required to bring down the temperature of the vessel and its contents to 5 °C.
Specific latent heat of fusion of ice = 336 × 103 J kg-1
Specific heat capacity of copper vessel = 400 J kg-1 °C-1
Specific heat capacity of water = 4200 J kg-1 °C-1.
