Advertisements
Advertisements
Question
The IUPAC name of \[\begin{array}{cc}
\ce{CH3}\phantom{.....................}\\
\backslash\phantom{....................}\\
\ce{CH - CH - COOH}\\/\phantom{.......}|\phantom{.............}\\
\ce{Br}\phantom{.......}\ce{CH3}\phantom{...........}\\\end{array}\] is
Options
2 – Bromo – 3- methyl butanoic acid
2 – methyl – 3 bromo butanoic acid
2 – methyl – 3 bromo butanoic acid
3 – Bromo – 2, 3 – dimethyl propanoic acid
Solution
2 – methyl – 3 bromo butanoic acid
APPEARS IN
RELATED QUESTIONS
Identify a more favourable resonance structure from the following. Justify.
\[\begin{array}{cc}
\phantom{.............}\ce{O-}\phantom{......................}\ce{O+}\\\phantom{............}|\phantom{.........................}|\\
\ce{^{+}CH3 - CH = C - H ↔ ^{-}CH2 - CH = C - H}
\end{array}\]
Observe the following structures and answer the questions given below.
- \[\ce{CH3 - CH2 - CH2 - CHO}\]
\[\begin{array}{cc}\ce{CH3 - CH - CHO}\\
|\phantom{...}\\\ce{CH3}\end{array}\]
a. What is the relation between (i) and (ii)?
b. Write IUPAC name of (ii).
c. Draw the functional group isomer of (i).
Phytane is a naturally occurring alkane produced by the alga spirogyra and is a constituent of petroleum. The IUPAC name for phytane is 2,6,10,14-tetramethylhexadecane. Write a zig-zag formula for phytane. How many primary, secondary, tertiary, and quaternary carbons are present in this molecule?
In the hydrocarbon
![]() the state of hybridisation of carbon 1,2,3,4 and 7 are in the following sequence.
the state of hybridisation of carbon 1,2,3,4 and 7 are in the following sequence.
Give the IUPAC names of the following compound.
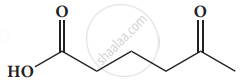
Give the IUPAC names of the following compound.
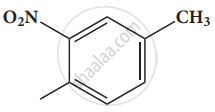
Give the structure for the following compound.
1,3,5- Trimethyl cyclohex - 1 -ene
Give the structure for the following compound.
1,3- Dimethylcyclohexane
Give the structure for the following compound.
3 - methylbutan - 2 - ol
The correct structure of 2,6-Dimethyl-dec-4-ene is ______
