Advertisements
Advertisements
Question
Phytane is a naturally occurring alkane produced by the alga spirogyra and is a constituent of petroleum. The IUPAC name for phytane is 2,6,10,14-tetramethylhexadecane. Write a zig-zag formula for phytane. How many primary, secondary, tertiary, and quaternary carbons are present in this molecule?
Solution
Zig-zag formula of phytane (2,6,10,14-tetramethylhexadecane) is as follows:

2,6,10,14-tetramethylhexadecane
Dash formula to represent types of C-atom:
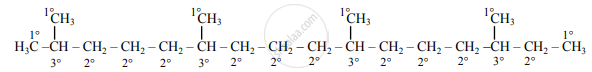
In phytane, six 1° C-atoms, ten 2° C-atoms, four 3° C-atoms are present. Phytane does not contain any quaternary carbon atom in its structure.
APPEARS IN
RELATED QUESTIONS
Identify a more favourable resonance structure from the following. Justify.

Match the pairs.
| Column 'A' | Column 'B' | ||
| i. | Inductive effect | a. | delocalisation of π electrons |
| ii. | Hyperconjugation | b. | displacement of π electrons |
| iii. | Resonance effect | c. | delocalisation of σ electrons |
| d. | displacement of σ electrons | ||
Write IUPAC names of the following.

Write the IUPAC name of the following.
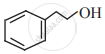
Write the IUPAC name of the following.

Observe the following and answer the questions given below:
\[\ce{CH3 - CH3 ->[U.V. light] \overset{\bullet}{C}H3 + \overset{\bullet}{C}H3}\]
- Name the reactive intermediate produced.
- Indicate the movement of electrons by a suitable arrow to produce this intermediate.
- Comment on the stability of this intermediate produced.
An electronic displacement in a covalent bond is represented by the following notation.
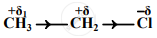
A. Identify the effect
B. Is the displacement of electrons in a covalent bond temporary or permanent.
In the hydrocarbon
![]() the state of hybridisation of carbon 1,2,3,4 and 7 are in the following sequence.
the state of hybridisation of carbon 1,2,3,4 and 7 are in the following sequence.
The IUPAC name of the compound is
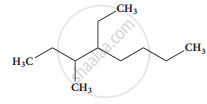
The IUPAC name of \[\begin{array}{cc}
\phantom{.}\ce{CH3}\\|\phantom{..}\\
\ce{H3C - C - CH = C(CH3)2}\\
|\phantom{..}\\\phantom{..}\ce{CH3}
\end{array}\] is
Give the IUPAC names of the following compound.
\[\begin{array}{cc}\ce{CH3 - CH2 - CH - CHO}\\
\phantom{.....}|\\\phantom{.......}\ce{OH}
\end{array}\]
Give the IUPAC names of the following compound.
\[\ce{CH2 = CH - CH = CH2}\]
Give the IUPAC names of the following compound.
\[\begin{array}{cc}\ce{CH3 - C ≡ C - CH - CH3}\\
\phantom{........}|\\\phantom{.........}\ce{Cl}
\end{array}\]
Give the IUPAC names of the following compound.
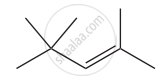
Give the IUPAC names of the following compound.
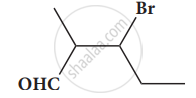
Give the structure for the following compound.
3 – ethyl – 2 methyl – 1 – pentene
As per IUPAC nomenclature, the name of the complex [Fe(H2O)5(NCS)]2+ is ____________.
As per IUPAC nomenclature, the name of the complex Na3[AlF6] is ____________.
\[\ce{CH3CH2OCH2CH2CH3}\] in IUPAC is known as ____________.
What is the IUPAC name of the following compound?
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH2 - CH - CH2 - CH2 - CH3}\\
\phantom{}|\phantom{.....}|\phantom{.............}|\phantom{...............}\\
\phantom{}\ce{CH3}\phantom{.}\ce{CH3}\phantom{..........}\ce{C2H5}\phantom{...........}
\end{array}\]
IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3 - C - CH2CH3}\\
|\phantom{....}\\
\ce{OH}\phantom{..}
\end{array}\]
The correct structure of 2,6-Dimethyl-dec-4-ene is ______
The IUPAC name of the following compound is:
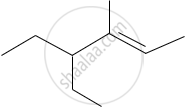
Identify the α-carbons in the following species and give the total number of α-hydrogen.
CH2 = CH - CH2 - CH3
Write the structural formulae for the following name and also write the correct IUPAC names for that.
2,2,3-trimethylpentan-4-ol
Write the structural formulae for the following name and also write the correct IUPAC name for that.
2,2,3-trimethylpentan-4-ol
