Advertisements
Advertisements
Question
What are the common types of secondary structure of proteins?
Solution 1
There are two common types of secondary structure of proteins:
(i) ∝-helix structure
(ii) β-pleated sheet structure
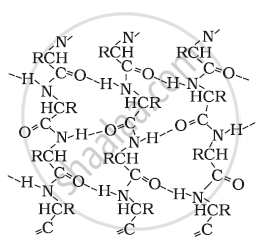
∝- Helix structure:
In this structure, the −NH group of an amino acid residue forms H-bond with the 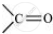 group of the adjacent turn of the right-handed screw (∝-helix).
group of the adjacent turn of the right-handed screw (∝-helix).

β-pleated sheet structure:
This structure is called so because it looks like the pleated folds of drapery. In this structure, all the peptide chains are stretched out to nearly the maximum extension and then laid side by side. These peptide chains are held together by intermolecular hydrogen bonds.
Solution 2
The conformation which the polypeptide chains assume as a result of hydrogen bonding is called secondary structure of the proteins. The two types of secondary structures are α-helix and β-pleated sheet structure.
APPEARS IN
RELATED QUESTIONS
Write one difference between α-helix and β-pleated structures of proteins.
The protein responsible for blood clotting is ____________.
The correct statement for protein haemoglobin.
Dinucleotide is obtained by joining two nucleotides together by phosphodiester linkage. Between which carbon atoms of pentose sugars of nucleotides are these linkages present?
Optical rotations of some compounds along with their structures are given below which of them have D configuration.
| (I) | 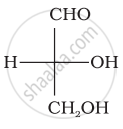 |
| (II) | 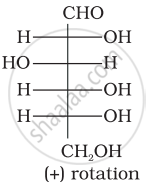 |
| (III) | 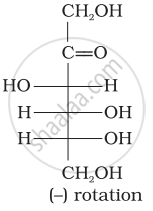 |
Proteins can be classified into two types on the basis of their molecular shape i.e., fibrous proteins and globular proteins. Examples of globular proteins are:
(i) Insulin
(ii) Keratin
(iii) Albumin
(iv) Myosin
Protein found in a biological system with a unique three-dimensional structure and biological activity is called a native protein. When a protein in its native form, is subjected to a physical change like change in temperature or a chemical change like, change in pH, denaturation of protein takes place. Explain the cause.
Which moieties of nucleosides are involved in the formation of phosphodiester linkages present in dinucleotides? What does the word diester in the name of linkage indicate? Which acid is involved in the formation of this linkage?
Assertion: β-glycosidic linkage is present in maltose,

Reason: Maltose is composed of two glucose units in which C–1 of one glucose unit is linked to C–4 of another glucose unit.
The main structural feature of proteins is
Each polypeptide in a protein has amino acids linked with each other in a specific sequence. This sequence of amino acids is said to be ______.
Explain formation of peptide linkage in protein with an example.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Proteins are found to have two different types of secondary structures viz alpha-helix and beta-pleated sheet structure.
Reason (R): The secondary structure of proteins is stabilized by hydrogen bonding.
Select the most appropriate answer from the options given below:
Out of the following, which type of interaction is responsible for the stabilisation of the α-helix structure of proteins?
The correct structure of Ruhemann's Purple, the compound formed in the reaction of ninhydrin with proteins is:
An α-helix is a structural feature of ______.
