Advertisements
Advertisements
Question
What do we observe on pouring acetic acid on red and blue litmus papers?
(A) Red litmus remains red and blue litmus turns red.
(B) Red litmus turns blue and blue litmus remains blue.
(C) Red litmus turns blue and blue litmus turns red.
(D) Red litmus becomes colourless and blue litmus remains blue.
Solution
(A) Red litmus remains red and blue litmus turns red.
Acids turn blue litmus paper red. They have no effect on red litmus paper.
APPEARS IN
RELATED QUESTIONS
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute sulphuric acid reacts with zinc granules.
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute hydrochloric acid reacts with magnesium ribbon.
Name one natural source of the following acids:
Lactic acid
Complete and balance the followingchemicalequations:
NaHCO3 (S) + HCI (aq) →
Explain why, sodium hydrogencarbonate is used as an antacid.
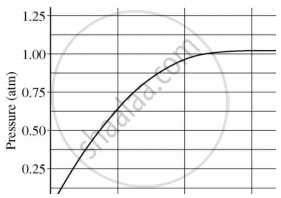
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
What happens when a solution of an acid is mixed with a solution of a base in a test tube?
- The temperature of the solution increases
- The temperature of the solution decreases
- The temperature of the solution remains the same
- Salt formation takes place
A salt may be ______.
What property do acids and bases have in common? Explain it with an example.
Some metals react with acids to produce salt and hydrogen gas. Illustrate it with an example. How will you test the presence of this gas?
