Advertisements
Advertisements
Question
What happens when benzene diazonium chloride is heated with water?
Solution
When benzene diazonium chloride is heated with water, it forms phenol along with nitrogen and hydrochloric acid as by-products as shown below:
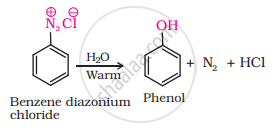
APPEARS IN
RELATED QUESTIONS
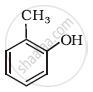
Write IUPAC name of the following compound:
Give IUPAC name of the following ether:
CH3CH2CH2OCH3
Write the structures of the products when Butan-2-ol reacts with SOCl2
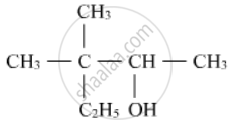
Write the IUPAC name of the following :
C6H5OCH2CH3 is called:
The IUPAC name of the ether CH2 = CH–CH2OCH3 is:
IUPAC name of m-cresol is ______.
Give IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - CH2 - CH2 - CH - CH3}\phantom{.}\\
\phantom{.........}|\phantom{...................}|\phantom{...........}\\
\phantom{..}\ce{Cl}\phantom{.................}\ce{OH}\phantom{..}
\end{array}\]
Explain why p-nitrophenol is more acidic than phenol.
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
