Advertisements
Advertisements
Question
What is meant by 'disproportionation'? Give two examples of disproportionation reaction in aqueous solution.
Solution
The oxidation and reduction of a substance at the same time as a result of a chemical reaction is called disproportionation. For example,
\[\ce{3\overset{0}{Cl2} + 6OH^- -> 5\overset{-1}{Cl}^- + \overset{+5}{Cl}O^-_3 + 3H2O}\]
\[\ce{3\overset{+6}{Mn}O^{2-}_4 + 4H^+ -> 2\overset{+7}{Mn}O^-_4 + \overset{+4}{Mn}O2 + 2H2O}\]
APPEARS IN
RELATED QUESTIONS
What are interstitial compounds?
Explain why is Fe3+ more stable than Fe2+?
Account for the following :
Zn is not considered as a transition element.
What may be the stable oxidation state of the transition element with the following d electron configurations in the ground state of their atoms?
3d3, 3d5, 3d8 and 3d4
Transition elements show magnetic moment due to spin and orbital motion of electrons. Which of the following metallic ions have almost same spin only magnetic moment?
(i) \[\ce{Co^{2+}}\]
(ii) \[\ce{Cr^{2+}}\]
(iii) \[\ce{Mn^{2+}}\]
(iv) \[\ce{Cr^{3+}}\]
Which of the following will not act as oxidising agents?
(i) \[\ce{CrO3}\]
(ii) \[\ce{MoO3}\]
(iii) \[\ce{WO3}\]
(iv) \[\ce{CrO^{2-}4}\]
Identify A to E and also explain the reactions involved.
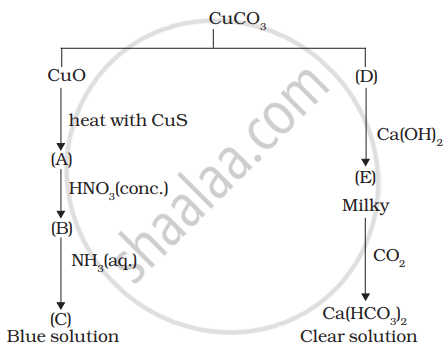
Identify the metal and justify your answer.
Carbonyl \[\ce{M(CO)5}\]
Catalytic hydrogenation of benzene gives
The standard electrode potentials of four elements A, B, C and D are – 3.05, – 1.66, – 0.40 and + 0.80. The highest chemical reactivity will be exhibited by
Which of the following statements is not correct?
Mercury is the only metal liquid at room temperature due to its:-
Which of the following ions will exhibit colour in aqueous solution?
Which of the following transition metal is not coloured?
Give reason for the following statement:
Physical and chemical properties of the 4d and 5d series of the transition elements are quite similar to expected.
A transition element X has an electronic configuration [Ar]4s23d3. Predict its likely oxidation states.
Account for the following:
Copper has an exceptionally positive `"E"_("M"^(2+)//"M")^0` value.
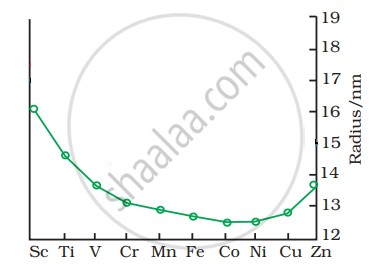
The trend of which property is represented by the following graph?
Give a reason for the following:
Transition metals possess a great tendency to form complex compounds.
