Advertisements
Advertisements
Question
What is the action of following on proponal?
Hydrazine
Solution
Propanal will undergo an addition elimination reaction with hydrazine to give corresponding hydrazone derivative containing C = N bond (imine).
\[\begin{array}{cc}
\phantom{}\ce{H}\phantom{....................................}\ce{H}\phantom{.......}\\
\phantom{}|\phantom{.....................................}|\phantom{........}\\
\ce{\underset{\text{Propanal}}{H3CH2C - C = O} + \underset{\text{Hydrazine}}{H2N - NH2}->\underset{\text{Hydrazone derivative}}{H3CH2C - C = N - NH2} + H2O}
\end{array}\]
RELATED QUESTIONS
Answer in brief.
Formic acid is stronger than acetic acid. Explain.
Write the structure of the product formed when carboxylic acid is heated with a dehydrating agent like P2O5
Write the preparation reactions for acid amide from the following.
Carboxylic acid
Write the preparation reactions for acid amide from the following.
Acid chloride
Write reactions for the following conversions.
Propanone to Propane
Write chemical reactions to convert –COOH group of acetic acid into the following.
CH4
Write chemical reactions to convert –COOH group of acetic acid into the following.
CH3COCl
Write reaction for preparation of acetophenone from benzoyl chloride.
Explain the acidic nature of carboxylic acids.
Write reactions for the action of the following reagents on p-chlorobenzaldehyde.
Ethane-1,2-diol in presence of dry HCl.
Complete the following sequence of reactions and write structures for A, B, C.
\[\ce{Dry ice ->[i. Dry ether][ii. Hydrolysis] A ->[PCl5] B ->[H2(gas)][Pd - BaSO4] C}\]
Which is the gas evolved when carboxylic acids react with strongly electropositive metals (such as Na, K, Ca, Zn)?
The product of the reaction between dialkyl cadmium and acyl chloride is a/an ____________.
In the following reaction:
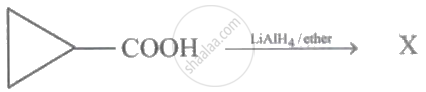
The product X is:
A flavouring agent found in oil of wintergreen is ______.
The elimination of CO2 from a carboxylic acid is known as ____________.
The reducing agent preferred to convert carboxylic acids and esters to primary alcohols is ____________.
The oxidation number of B in NaBH4 is ____________.
Which of the following functional groups is reduced by diborane?
Which of the following compounds is obtained when ethanoic anhydride is treated with water?
Which of the following carboxylic acids ha highest acidic strength?
Which oxide of nitrogen is obtained on heating ammonium nitrate at 250°C?
Excess of ammonia with sodium hypochloride solution in the presence of glue or gelatine gives ______.
Which of the following can reduce?
\[\ce{RCOOH -> RCH2OH}\]
Draw structures 01 conjugate bases of monochloroacetic acid and dichloroacetic acid. Which one is more stabilized by -I effect?
