Advertisements
Advertisements
Question
Answer in brief.
Formic acid is stronger than acetic acid. Explain.
Solution
- The negatively charged acetate ion (i.e., the conjugate base of acetic acid) gets destabilized by + I effect of – CH3 group.
- Lesser is the stabilization of the conjugate base, weaker is the acid.
- In formate ion, there is no such destabilization effect.
Thus, formic acid is stronger than acetic acid.
APPEARS IN
RELATED QUESTIONS
Arrange the following carboxylic acids with increasing order of their acidic strength and justify your answer.
1) ![]()
2) ![]()
3) ![]()
Correct order of acid strength for
- acetic acid
- fluoroacetic acid
- 4-Nitrobenzoic acid
- 4-Methyl benzoic acid is ______.
Write the structure of the product formed when carboxylic acid is heated with a dehydrating agent like P2O5
What is the action of following on proponal?
Hydrazine
Write the preparation reactions for acid amide from the following.
Carboxylic acid
Write reactions for the following conversions.
Propanone to Propane
Write reactions for the following conversions.
4-Nitrobenzoic acid to Nitrobenzene
Write chemical reactions to convert –COOH group of acetic acid into the following.
CH4
Write chemical reactions to convert –COOH group of acetic acid into the following.
CH3COCl
Write reaction for preparation of acetophenone from benzoyl chloride.
Write reactions for the action of the following reagents on p-chlorobenzaldehyde.
Ethane-1,2-diol in presence of dry HCl.
Which of the following compounds reacts with ammonia to form urotropine?
In the reaction, \[\ce{C6H5COCH3 ->[{[H]}][Zn-Hg/conc. HCl] X}\], X is ______.
Which is the gas evolved when carboxylic acids react with strongly electropositive metals (such as Na, K, Ca, Zn)?
Which of following elements does not form amide when reacted with ammonia?
In the following reaction:
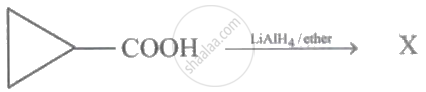
The product X is:
A flavouring agent found in oil of wintergreen is ______.
Which of the following carboxylic acids will have the highest acidity?
The elimination of CO2 from a carboxylic acid is known as ____________.
Which among the following is the strongest acid?
The oxidation number of B in NaBH4 is ____________.
Which of the following aromatic acids has less acidic strength than benzoic acid?
Which of the following compounds is obtained when ethanoic anhydride is treated with water?
Which oxide of nitrogen is obtained on heating ammonium nitrate at 250°C?
The number of bonding pairs and lone pairs of electrons present in the central atom of ammonia molecular are respectively.
