Advertisements
Advertisements
Question
Which of the following species has tetrahedral geometry?
Options
\[\ce{BH^{-}4}\]
\[\ce{NH^{-}2}\]
\[\ce{CO^{2-}3}\]
\[\ce{H3O+}\]
Solution
\[\ce{BH^{-}4}\]
Explanation:
(i) The molecule of \[\ce{BH^{−}4}\] has four bond pairs and zero lone pair of electrons, so it will be a tetrahedral molecule.
(ii) \[\ce{NH^{-}2}\] has two bond pairs and two lone pairs of electrons on the nitrogen atom. So, it will have a bent geometry.
(iii) \[\ce{CO^{2-}3}\] has three bond pairs and no lone pairs of electrons on carbon atoms. So, it will have a trigonal planar geometry.
(iv) \[\ce{H3O+}\] has three bond pairs and one lone pair of electrons on oxygen atoms. So, it has a pyramidal geometry.
APPEARS IN
RELATED QUESTIONS
Explain the important aspects of resonance with reference to the `"CO"_3^(2-)` ion.
H3PO3 can be represented by structures 1 and 2 shown below. Can these two structures be taken as the canonical forms of the resonance hybrid representing H3PO3? If not, give reasons for the same.
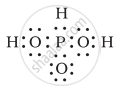 |
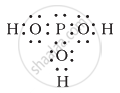 |
| (1) | (2) |
Write the resonance structure for SO3.
Write the resonance structures for NO2.
Write the resonance structures for `"NO"_3^(-)`.
Explain why \[\ce{CO^{2–}3}\] ion cannot be represented by a single Lewis structure. How can it be best represented?
Draw the resonating structure of ozone molecule
Draw the resonating structure of nitrate ion
Resonance structures can be written for ______.
The acceptable resonating structures of the following molecule are:
\[\begin{array}{cc}
\ce{CH3 - CH = C - \overset{\bullet\bullet}{\underset{\bullet\bullet}{O}} - CH2 - CH3}\\
|\phantom{......}\\
\ce{:N}\phantom{.......}\\
\phantom{}/\phantom{...}\backslash\phantom{......}\\
\phantom{}\ce{H3C}\phantom{.....}\ce{CH3}\phantom{......}
\end{array}\]
| (x) | \[\begin{array}{cc} \ce{CH3 - \overset{\overset{Θ}{\bullet\bullet}}{C}H - C - \overset{\bullet\bullet}{\underset{\bullet\bullet}{O}} - CH2 - CH3}\\ ||\phantom{......}\\ \ce{N^⊕}\phantom{.....}\\ \phantom{}/\phantom{...}\backslash\phantom{......}\\ \phantom{}\ce{H3C}\phantom{.....}\ce{CH3}\phantom{......} \end{array}\] |
| (y) | \[\begin{array}{cc} \ce{CH3 - \overset{\overset{Θ}{\bullet\bullet}}{C}H - C = \overset{⊕}{\underset{\bullet\bullet}{O}} - CH2 - CH3}\\ |\phantom{......}\\ \ce{:N}\phantom{.......}\\ \phantom{}/\phantom{...}\backslash\phantom{......}\\ \phantom{}\ce{H3C}\phantom{.....}\ce{CH3}\phantom{......} \end{array}\] |
| (z) | \[\begin{array}{cc} \ce{CH3 - \overset{⊕}{C}H - C - \overset{\bullet\bullet}{\underset{\bullet\bullet}{O}} - CH2 - CH3}\\ ||\phantom{......}\\ \ce{N^Θ}\phantom{.....}\\ \phantom{}/\phantom{...}\backslash\phantom{......}\\ \phantom{}\ce{H3C}\phantom{.....}\ce{CH3}\phantom{......} \end{array}\] |
| (w) | \[\begin{array}{cc} \ce{CH3 - \overset{⊕}{C}H - C = \overset{Θ}{O} - CH2 - CH3}\\ |\phantom{......}\\ \ce{:N}\phantom{.......}\\ \phantom{}/\phantom{...}\backslash\phantom{......}\\ \phantom{}\ce{H3C}\phantom{.....}\ce{CH3}\phantom{......} \end{array}\] |
