Advertisements
Advertisements
Question
Why are certain compounds called hydrocarbons? Write the general formula for homologous series of alkanes, alkenes and alkynes and also draw the structure of the first member of each series. Write the name of the reaction that converts alkenes into alkanes and also write a chemical equation to show the necessary conditions for the reaction to occur.
Solution
The compounds entirely consisting of carbons and hydrogens are known as Hydrocarbons. There are different categories in which hydrocarbons are divided out of which the two are:
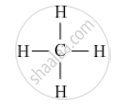
Saturated Hydrocarbons: The compounds of carbon having only single bonds between the carbon atoms are called saturated compounds. This includes alkanes, having a general formula CnH2n+2.The first member of homologous series of alkanes is methane (CH4).
Structure of methane is as follows:
Unsaturated Hydrocarbons: The compounds of carbon having double and triple bonds between the carbon atoms are called unsaturated compounds. This includes alkenes and alkynes having general formula CnH2n and CnH2n-2, respectively. The first member of homologous series of alkenes is Ethene (C2H4).
The structural formula of ethene is as follows: H2C=CH2.
The first member of homologous series of alkynes is Ethyne (C2H2) having structural formula: HC=CH.
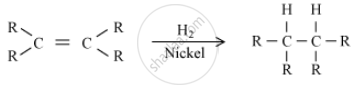
The reaction which converts unsaturated hydrocarbons to saturated hydrocarbons, i.e. alkenes to alkanes is know as Hydrogenation Reaction.
It is used to obtain ghee from oil. Vegetable oil contains unsaturated fatty acids, and is liquid at room temperature. When they are hydrogenated, they become saturated and change to a solid state.
The reaction is as follows:
Hydrogen can be added to unsaturated hydrocarbons in the presence of nickel to yield saturated hydrocarbons. In this reaction, nickel acts as a catalyst.
APPEARS IN
RELATED QUESTIONS
Give any two differences between alkanes and alkenes.
Define Saturated hydrocarbon.
A compound 'X' on heating with excess conc. sulphuric acid at 443 K gives an unsaturated compound 'Y'. 'X' also reacts with sodium metal to evolve a colourless gas 'Z'. Identify 'X', 'Y' and 'Z'. Write the equation of the chemical reaction of formation of 'Y' and also write the role of sulphuric acid in the reaction.
Write a short note on Catenation.
Write the electron-dot structures for ethene.
Giving their structures, state the number of single bonds, double bonds and triple bonds (if any) in the following compounds:
ethyne
Answer the following question.
Name a cyclic unsaturated carbon compound.
The correct electron dot structure of a water molecule is
Name the functional groups present in the following compounds
- CH3 CO CH2 CH2 CH2 CH3
- CH3 CH2 CH2 COOH
- CH3 CH2 CH2 CH2 CHO
- CH3 CH2 OH
Carbon, Group (14) element in the Periodic Table, is known to form compounds with many elements. Write an example of a compound formed with
- chlorine (Group 17 of Periodic Table)
- oxygen (Group 16 of Periodic Table)
