Advertisements
Advertisements
Question
Write an equation to illustrate the reducing nature of ammonia.
Solution
\[\ce{8NH3(excsess) + 3CI2 -> N2 + 6NH4CI}\]
APPEARS IN
RELATED QUESTIONS
What property of ammonia is demonstrated by the fountain experiment?
Ammonium salts decompose on heating. What other property do ammonium salts have in common?
State what you observe when a piece of moist red litmus paper is placed in a gas jar of ammonia.
Describe two tests to identify ammonia and ammonium ions in an aqueous solution.
State your observation for the following cases
(i) Ammonia gas is burnt in an atomosphere of oxygen in the absence of a catalyst
(ii) Glass rod dipped in ammonium hydroxide is brought near the mouth of the concentrated hydrochloric acid bottle
The diagram shows a simple arrangement of the fountain experiment:
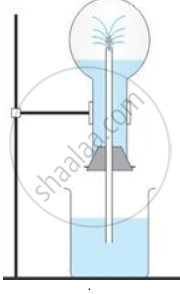
(i) Name the two gases you have studied which can be used in this experiment
(ii) What is the common properly demonstrated by this experiment
The diagram shows an experimental set up for the laboratory preparation of a pungent smelling gas. The gas is alkaline in nature.

- Nature the gas collected in the jar.
- Write the balance equation for the above preparation.
- How is the gas being collected?
- Name the drying agent used.
- How will you find that the jar is full of gas?
What do you observe when Ammonium solution is added to ferric chloride solution.
