Advertisements
Advertisements
Question
Write preparation of low density polythene.
Solution
Preparation of low density polythene:
- LDP is prepared by polymerization of ethylene under high pressure (1000 – 2000 atm) and temperature (350 – 570 K) in presence of traces of O2 or peroxide as initiator.
\[\ce{{n} CH2 = CH2 ->[Traces of O2 or][peroxide at 350–570 K, 1000–2000 atm] LDP}\] - The mechanism of this reaction involves free radical addition and H-atom abstraction. The latter results in branching.
- Polymeric chains are loosely held due to branching and the polymer has low density.
APPEARS IN
RELATED QUESTIONS
Bakelite is the polymer of:
(a) Benzaldchyde and phenol
(b) Acetaldehyde and phenol
(c) Formaldehyde and phenol
(d) Formaldehyde and benzyl alcohol
Based on molecular forces, what type of polymer is neoprene?
Write the formulae of the raw materials used for preparation of Dextran.
Answer the following in one sentence.
Define the term: Vulcanization
Answer the following in one sentence.
Identify thermoplastic and thermosetting plastic from the following:
- PET
- Urea formaldehyde resin
- Polythene
- Phenol formaldehyde resin
Answer the following.
Write the reaction of the formation of Terylene.
Answer the following.
Match the following pairs:
| Name of polymer | Monomer |
| 1. Teflon | a. CH2 = CH2 |
| 2. PVC | b. CF2 = CF2 |
| 3. Polyester | c. CH2 = CHCl |
| 4. Polythene | d. C6H5OH and HCHO |
| 5. Bakelite | e. Dicarboxylic acid and polyhydoxyglycol |
Draw the structures of polymers formed from the following monomers
\[\ce{n HOOC–R–COOH + n HO–R'–OH}\]
Name and draw structure of the repeating unit in natural rubber.
Identify condensation polymers and addition polymers from the following.
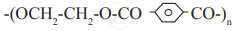
Attempt the following:
Write the chemical reactions involved in the manufacture of Nylon 6,6.
Answer the following.
Write main specialities of Buna-S, Neoprene rubber?
Mention two uses of LDP.
Write chemical reaction for preparation of the following.
Buna-S
Write the name and formulae of the monomers used for the preparation of dacron.
Write chemical reactions for the preparation of high-density polythene.
Define rubber.
Which among the following polymers is obtained from styrene and 1-3-butadiene?
Which among the following polymers is used for making handles of cooker?
Novolac is obtained from ____________.
Which of the following catalysts is used in preparation of terylene?
Which among the following catalysts is used in the preparation of dacron?
Which among the following monomers is used to prepare Teflon?
Which of the following monomers is used in the manufacture of Neoprene rubber?
Which of the following pair of compounds is used as monomers for bakelite?
Which of the following polymer is used to make blankets?
Which among the following is an example of addition polymer?
Which of the following polymers, need atleast one diene monomer for their preparation?
(i) Dacron
(ii) Buna-S
(iii) Neoprene
(iv) Novolac
Match the polymers given in Column I with the type of linkage present in them given in Column II.
| Column I | Column II |
| (i) Terylene | (a) Glycosidic linkage |
| (ii) Nylon | (b) Ester linkage |
| (iii) Cellulose | (c) Phosphodiester linkage |
| (iv) Protein | (d) Amide linkage |
| v) RNA |
Phenol and formaldehyde undergo condensation to give a polymar (A) which on heating with formaldehyde gives a thermosetting polymer (B). Name the polymers. Write the reactions involved in the formation of (A). What is the structural difference between two polymers?
F2C = CF2 is monomer of the polymer -
Trans - form of poly isoprene is:-
Which of the following is an example of polyester?
Which of the following polymers is synthesized using a free radical polymerisation technique?
Polymer used in bullet proof glass is ______.
Which of the following is a polymer of enzyme?
Which of the foolowing polymer is used in the manufacture of insulators.
The monomer of natural rubber is ______.
Answer the following.
Name and draw structure of the repeating unit in natural rubber.
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the name of the monomer of natural rubber.
Another name of terylene is ______.
The monomer used in preparation of teflon is ______.
Write the structure and name of monomer of Natural rubber.
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
