Advertisements
Advertisements
Question
Write the name and formulae of the monomers used for the preparation of dacron.
Solution
Monomers used in the preparation of dacron are ethylene glycol and terephthalic acid.
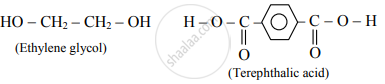
APPEARS IN
RELATED QUESTIONS
Write any ‘two' uses of terylene.
Based on molecular forces, what type of polymer is neoprene?
The Zieglar-Natta catalyst is used in the preparation of _______.
(A) LDPE
(B) PHBV
(C) PAN
(D) HDPE
Answer the following in one sentence.
Define the term: Vulcanization
Identify condensation polymers and addition polymers from the following.
\[\begin{array}{cc}\ce{-(CH2 - CH -)_{n}}\\
\phantom{....}|\\\ce{\phantom{.......}C6H5}
\end{array}\]
Identify condensation polymers and addition polymers from the following.
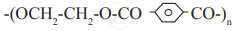
Attempt the following:
Explain the vulcanisation of rubber. Which vulcanizing agents are used for the following synthetic rubber?
a. Neoprene
b. Buna-N
Attempt the following:
What is meant by LDP and HDP? Mention the basic difference between the same with suitable examples.
Attempt the following:
Write preparation, properties and uses of Teflon.
Answer the following.
Is synthetic rubber better than natural rubber? If so, in what respect?
Answer the following.
Write main specialities of Buna-S, Neoprene rubber?
Write the structure of isoprene and the polymer obtained from it.
Write the name of the catalyst used for preparation of high density polythene polymer.
Monomers ethylene glycol and terephthalic acid undergo condensation polymerization to give polymer called ___________
Monomer of natural rubber is __________
Write the name of one example of each polymer in which following repeating units.
\[\begin{array}{cc}
\ce{(-CF2-CF2-), -[NH-(CH2)5-CO] -, -(CH2-CH-), (-CH2-CH2-)}\\
\phantom{............................}|\\
\phantom{..............................}\ce{CN}
\end{array}\]
Which of the following polymer is used in paints?
Identify the CORRECT statement regarding the following polymer.
\[\begin{array}{cc}
\phantom{....}\ce{O}\phantom{............}\ce{O}\phantom{...................}\ce{H}\phantom{.....}\\
\phantom{....}||\phantom{.............}||\phantom{...................}|\phantom{......}\\
\ce{-[C - (CH2)4 - C - NH - (CH2)6 - N -]_{{n}}}
\end{array}\]
Identify the INCORRECT match.
Which of the following polymers is obtained from chloroprene?
Which of the following polymers is a heteropolymer?
Identify the polymer obtained by polymerization of n moles of acrylonitrile.
Which among the following polymers can NOT be remoulded?
Which of the following compounds is used to prepare orlon?
Which among the following polymers is an example of addition polymer?
Which of the following pair of compounds is used as monomers for bakelite?
Name the polymers used in laminated sheets and give the name of monomeric units involved in its formation.
Trans - form of poly isoprene is:-
Nylon threads are made of ______.
The catalyst used for the polymerisation of olefins is ______.
Which among the following polymers has high tensile strength and is used to obtain tyre cords?
Which of the following is a polymer of enzyme?
Which of the foolowing polymer is used in the manufacture of insulators.
The monomer of natural rubber is ______.
Answer the following.
Name and draw structure of the repeating unit in natural rubber.
Answer the following.
Write the structure of isoprene and the polymer obtained from it.
Answer the following.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name the compound which reacts with formaldehyde to produce ethyl alcohol.
Write the preparation of viscose rayon.
Write the name of the monomer of natural rubber.
Another name of terylene is ______.
Match the following pairs:
| Polymer | Monomer | ||
| (i) | Teflon | (a) | CH2 = CH2 |
| (ii) | PVC | (b) | CF2 = CF2 |
| (iii) | Natural rubber | (c) | Isoprene |
| (iv) | Polythene | (d) | H2C=CHCl |
The monomer used in preparation of teflon is ______.
Write the structure and name of monomer of Nylon-6.
Write the structure and name of monomer of Natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
