Advertisements
Advertisements
Question
Write the reaction involved in the Stephen reduction
Solution
Stephen reduction: In this reaction, nitriles are reduced to corresponding imine with stannous chloride in the presence of hydrochloric acid, which on hydrolysis give corresponding aldehyde.
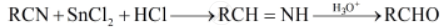
APPEARS IN
RELATED QUESTIONS
Write the product in the following reaction
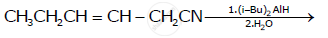
How will you bring about the following conversion?
Benzoyl chloride to benzaldehyde
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
p-Nitrobenzaldehyde
Esters react with DIBAL-H to produce:
Aldehydes are produced on reduction of the following by DIBAL-H:
The oxidation of toluene to benzoic acid can be stopped at the aldehyde stage to give benzaldehyde. The reagent used for the purpose is one of the following.
Aldehydes are prepared by reducing nitriles to corresponding imines with stannous chloride in the presence of hydrochloric acid. This reaction is called:
The oxidation of toluene to benzoic acid can be done using which of the following reagents.
Explain the following reactions:
Stephan reaction
Predict the reagent for carrying out the following transformations:
Benzoyl chloride to Benzaldehyde
