Advertisements
Advertisements
प्रश्न
Write the reaction involved in the Stephen reduction
उत्तर
Stephen reduction: In this reaction, nitriles are reduced to corresponding imine with stannous chloride in the presence of hydrochloric acid, which on hydrolysis give corresponding aldehyde.
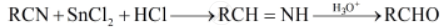
APPEARS IN
संबंधित प्रश्न
Write the product in the following reaction
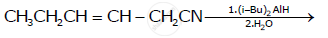
How will you bring about the following conversion?
Benzoyl chloride to benzaldehyde
When 0.4g of oxalic acid is dissolved in 40g of benzene, the freezing point of the solution is lowered by 0.45 K. Calculate the degree of association of acetic acid. Acetic acid forms dimer when dissolved in benzene.
(Kf for benzene = 5.12 K kg mol-1, at. wt. C = 12, H = 1, O = 16)
Aldehydes are prepared by reducing nitriles to corresponding imines with stannous chloride in the presence of hydrochloric acid. This reaction is called:
Benz aldehyde + NaOH →
Aldehydes are the first oxidation products of ______.
Predict the reagent for carrying out the following transformations:
Benzoyl chloride to Benzaldehyde
Assertion (A): Strong oxidising agents oxidise toluene and its derivatives to benzoic acids.
Reason (R): It is possible to stop the oxidation of toluene at the aldehyde stage with suitable reagents.
Select the most appropriate answer from the options given below:
An organic compound with molecular formula \[\ce{C7H7NO2}\] exists in three isomeric forms, the isomer ‘A’ has the highest melting point of the three. ‘A’ on reduction gives compound ‘B’ with molecular formula \[\ce{C7H9N}\]. ‘B’ on treatment with \[\ce{NaNO2/HCl}\] at 0-5° C to form compound ‘C’. On treating C with \[\ce{H3PO2}\], it gets converted to D with formula \[\ce{C7H8}\], which on further reaction with \[\ce{CrO2Cl2}\] followed by hydrolysis forms ‘E’ \[\ce{C7H6O}\]. Write the structure of compounds A to E. Write the chemical equations involved.
Account for the following:
N-ethylbenzene sulphonyl amide is soluble in alkali.
