Advertisements
Advertisements
Question
Write the correct order of increasing ease of dehydrohalogenation.
\[\ce{\underset{\text{(I)}}{CH3 - CH2 - CH2 - Cl}}\]
\[\ce{\underset{\text{(II)}}{CH3 - CH(Cl) - CH3}}\]
\[\begin{array}{cc}
\ce{CH3}\\
|\\
\ce{CH3-C-Cl}\phantom{...}\\
|\\
\ce{CH3}\\
\ce{(III)}
\end{array}\]
Solution
The correct order of increasing ease of dehydrohalogenation is, III > II > I.
APPEARS IN
RELATED QUESTIONS
Complete the following reaction giving major products.
\[\begin{array}{cc}\ce{CH3 - CH - CH3 ->[Red P/Br2] A ->[Ag2O/H2O]B}\\|\phantom{.........................}\\
\ce{OH\phantom{.......................}}\end{array}\]
Name the reagent used to bring about the following conversion.
Ethyl bromide to ethyl isocyanide
Give reasons:
Haloarenes are less reactive than haloalkanes.
In alkaline hydrolysis of tertiary butyl bromide, ____________.
Which of the following is least reactive towards nucleophilic addition?
The following mechanism has been proposed for the reaction of NO2 with F2 to form NO2F:
\[\ce{NO2_{(g)} + F2_{(g)} -> NO2F_{(g)} + F_{(g)} (slow)}\]
\[\ce{F_{(g)} + NO2_{(g)} -> NO2F_{(g)} (fast)}\]
The order of the reaction with respect to NO2(g)
\[\ce{C6H5CH2Cl + KCN(alc) -> X + Y}\]
Compounds X and Y are ____________.
When C2H5Br is treated with excess of alc. NH3, the major product obtained is ____________.
What is the major product obtained in the sulphonation of chlorobenzene with concentrated sulphuric acid?
The major product formed when 2-bromobutane is treated with alcoholic KOH is ______.
Which of the following statements is incorrect regarding the dehydrohalogenation of alkenes?
With which halogen the reactions, of alkanes are explosive?
Complete the following reactions giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Observe the following and answer the question given below:
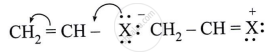
Can react by SN1 mechanism? Justify your answer.
Complete the following reaction sequences by writing the structural formulae of the organic compounds 'A', 'B' and 'C'.
\[\ce{2-Bromobutan ->[alc. KOH] A ->[][Br2] B ->[][NANH2] C}\]
Define and explain the SN1 mechanism with a suitable example.
Complete the reaction:
\[\ce{CH3CH2Cl ->[AgCN][alc.\Delta]}\] ?
Complete the following reaction, giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Identify the chiral molecule from the following.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/ dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}\\
\end{array}\]
