HSC Science (General)
HSC Science (Electronics)
HSC Science (Computer Science)
Academic Year: 2017-2018
Date: July 2018
Advertisements
- All questions are compulsory.
- Answers of both the sections should be written in same answer book.
- Draw well labelled diagrams and write balanced equations wherever necessary.
- Figures to the right indicate full marks.
- Use of logarithmic table is allowed.
- Every new question must be started on a new page.
What is the role of the following compound -
CaF2 in metallurgy of aluminum?
Chapter: [0.01] Solid State
What is the role of the following compound-
SiO2 in the extraction of copper from copper pyrites?
Chapter: [0.06] General Principles and Processes of Isolation of Elements
Classify the following molecular solids into different types:
- HCl
- CO2
- Solid ice
- SO2
Chapter: [0.01] Solid State [0.01] Solid State
Define the following terms :
a. Cryoscopic constant
b. Resistivity
Chapter: [0.02] Solutions and Colligative Properties
Arrange the following oxyacids of chlorine in decreasing order of their thermal stability. Give reason.
HOClO, HOCl, HClO4, HOClO2
Chapter: [7.03] Group 17 Elements
Derive van’t Hoff general solution equation
Chapter: [0.02] Solutions and Colligative Properties
Represent a cell consisting of Mg2+ | Mg half cell and Ag+
| Ag half cell and write the cell reaction.
( `"E"_("Ag")^° = 0.799 "V", "E"_("Mg")^° = - 2.37 "V"`)
Chapter: [0.01] Solid State
Find the overall order of the following reactions:
1. CHCl3(g) + Cl2(g) → CCl4(g) + HCl(g), Rate = k[CHCl3][Cl2]
2. 2NO(g) + O2(g) → 2NO2(g), Rate = k[NO]2[O2]
Chapter:
State and explain Hess’s law of constant heat summation.
Chapter: [0.03] Chemical Thermodynamics and Energetic
Explain the following with the help of balanced chemical equation:
a. Bleaching action of SO2.
b. Dehydration of formic acid by concentrated H2SO4.
c. Burning of benzene in presence of excess of dioxygen.
Chapter:
The density of silver having an atomic mass of 107.8 g mol- 1 is 10.8 g cm-3. If the edge length of cubic unit cell is 4.05 × 10- 8
cm, find the number of silver atoms in the unit cell.
( NA = 6.022 × 1023, 1 Å = 10-8 cm)
Chapter: [0.01] Solid State
How much quantity of electricity in coulomb is required to deposit 1.346 × 10-3 kg of Ag in 3.5 minutes from AgNO3 solution?
( Given: Molar mass of Ag is 108 × 10-3 kg mol-1 )
Chapter: [0.04] Electrochemistry
5 moles of helium expand isothermally and reversibly from a pressure 40 × 10-5 N m-2 to 4 × 10-5 N m-2 at 300 K. Calculate the work done, change in internal energy and heat absorbed during the expansion. (R = 8.314 J K-1 mol-1).
Chapter: [0.03] Chemical Thermodynamics and Energetic
Write structure and molecular formula for the following compounds:
a. Orthophosphoric acid
b. Sulphurous acid
Chapter: [7.02] Group 16 Elements
What is the ‘Ellingham diagram’? Write any ‘two points’ of its significance.
Chapter: [0.04] Electrochemistry
In a first-order reaction A → product, 80% of the given sample of compound decomposes in 40 min. What is the half-life period of the reaction?
Chapter: [0.05] Chemical Kinetics [0.06] Chemical Kinetics
Write features of a reversible process.
Chapter: [0.03] Chemical Thermodynamics and Energetic
Advertisements
Write any ‘four points’ of difference between fluorine and other halogens.
Chapter: [0.07] Elements of Groups 16, 17 and 18 [7.03] Group 17 Elements
What is the freezing point of a liquid? The freezing point of pure benzene is 278.4 K. Calculate the freezing point of the solution when 2.0 g of a solute having molecular weight 100 g mol-1
is added to 100 g of benzene.
( Kf of benzene = 5.12 K kg mol-1 .)
Chapter: [0.02] Solutions and Colligative Properties
A substance which shows the highest entropy is ___________.
SrCO3(s)
Cu(s)
NaCl(aq.)
Cl2(g)
Chapter: [0.03] Chemical Thermodynamics and Energetic
The process of extracting a soluble material from an insoluble solid by dissolving out in a suitable solvent is known as _________.
calcination
roasting
leaching
smelting
Chapter: [0.16] Chemistry in Everyday Life
The kinetic order for the following reaction is ________.
\[\ce{2N_2O_(g) ->[Pt] 2N_2_(g) + O_2_(g) }\]
zero
first
second
third
Chapter:
Which of the following compounds of chlorine is used as refrigerant?
CCl3NO2
CCl2F2
COCl2
CCl4
Chapter: [0.07] Elements of Groups 16, 17 and 18 [7.03] Group 17 Elements
4.0 grams of NaOH ( Molar mass = 40.0 g mol-1 ) is dissolved in 500 cm3 of water. What is the molarity of NaOH solution?
1 M
0.8 M
0.5 M
0.2 M
Chapter: [0.02] Solutions and Colligative Properties
Number of types of orthorhombic unit cell is ___________.
7
3
4
2
Chapter: [0.01] Solid State
The S.I. unit of cell constant for conductivity cell is __________.
m-1
S m-2
cm-2
S dm2 mol-1
Chapter: [0.04] Electrochemistry
Explain the geometry of [Cu(NH3)4]2+ on the basis of hybridisation [ At. No. Cu = 29 ].
Chapter: [7.01] Group 15 Elements
What are 'd' and 'f' block elements?
Chapter: [8.01] D-block Elements
Write a short note on Sandmeyer’s reaction.
Chapter: [10.01] Haloalkanes
What is the action of the following on isopropyl methyl ether?
a. Cold HI
b. Hot HI
Chapter: [16.01] Chemicals in Medicines
Write balanced equation for the following conversion:
a. Cyclopropane carboxylic acid to cyclopropylmethanol.
b. Acetyl chloride to benzyl methyl ketone using dialkyl cadmium.
Chapter: [12.02] Carboxylic Acids
What is the action of lithium aluminum hydride in the presence of ether on the following compounds?
a. Nitroethane
b. 2-Methyl-1-nitropropane
Chapter: [0.09] Coordination Compounds [0.09] Coordination Compounds
How is glucose prepared from starch?
Chapter: [14.01] Carbohydrates
Define:
a. Analgesics
b. Antimicrobials
Chapter: [0.16] Chemistry in Everyday Life
Advertisements
Complete and rewrite the balanced chemical equation for the following reactions:
a. \[\ce{1-chloro-2, 4-dinitrobenzene ->[aq. alkali][368 K, H+]}\] ?
b. \[\ce{Benzaldehyde ->[50‰ KOH] }\] ?
c. \[\ce{Acetone + phenylhydrazine ->[H+]}\] ?
Chapter:
Write a preparation of phenol from cumene? What happens when phenol is heated with zinc dust?
Chapter: [11.02] Phenols
What are racemates?
Chapter: [0.1] Halogen Derivatives [10.01] Haloalkanes
What is the action of following reagents on glucose?
1. Bromine water
2. Hydroxylamine
Chapter: [12.02] Carboxylic Acids
Distinguish between thermoplastic polymers and thermosetting polymers.
Chapter: [0.15] Polymers
What is the action of the following on lanthanoids?
a. water
b. Sulphur, heat
c. nitrogen, heat
Chapter: [8.02] F-block Elements
Draw the structures of veronal and thymine.
Chapter: [0.15] Introduction to Polymer Chemistry [0.15] Polymers
Identify A and B from the following reaction and rewrite complete reaction: 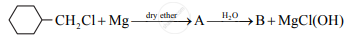
Chapter: [12.02] Carboxylic Acids
Write the preparation of benzoic acid from the following:
a. styrene
b. benzamide
c. dry ice
Chapter:
Write resonance structures of aniline. What is the action of benzene diazonium chloride on ethanol?
Chapter: [11.01] Alcohols
Write the formula for pentaamminechlorocobalt (III) sulphate.
Chapter: [7.02] Group 16 Elements
A polymer which contains ester linkage is_______.
teflon
buna-N
dextron
neoprene
Chapter:
An antihistamine drug is _______.
salvarsan
chloramphenicol
seldane
prontosil
Chapter: [0.16] Chemistry in Everyday Life
Identify A and B respectively in the following reaction:
\[\ce{tert-butyl alcohol ->[20‰H2SO4][363K] A + H2O ->[HBr][H2O2] B}\]
2 - Methylpropene, 1- bromo - 2 - methylpropane
2 - Methylpropene, 2 - bromo - 2 - methylpropane
2 - Methylpropane, 1 - bromo - 2 - methylpropane
2 - Methylpropane, 2 - bromo - 2 - methylpropane
Chapter: [11.01] Alcohols
The ligand triethylenetetramine is _______.
monodentate
bidentate
tridentate
tetradentate
Chapter: [0.09] Coordination Compounds
Pyrolusite ore is _______.
MnO
MnO2
Mn2O7
Mn2O3
Chapter: [0.01] Solid State
\[\ce{C2H5 - Br + Nal ->[dry acetone] C2H5 - I + NaBr}\]
The above reaction is _______.
Wurtz reaction
Balz-Schiemann reaction
Swarts reaction
Finkelstein reaction
Chapter:
The functional group present in triacylglycerol is _______.
alcohol
ether
ester
amine
Chapter: [12.02] Carboxylic Acids
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
Maharashtra State Board previous year question papers 12th Standard Board Exam Chemistry with solutions 2017 - 2018
Previous year Question paper for Maharashtra State Board 12th Standard Board Exam Chemistry-2018 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of Maharashtra State Board 12th Standard Board Exam.
How Maharashtra State Board 12th Standard Board Exam Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
