Topics
Some Basic Concepts of Chemistry
Introduction to Analytical Chemistry
- Introduction of Analytical Chemistry
- Analysis
- Mathematical Operation and Error Analysis
- Determination of Molecular Formula
- Chemical Reactions and Stoichiometric Calculations
- Limiting Reagent
- Concentration of a Solution
- Use of Graph in Analysis
Basic Analytical Techniques
- Introduction of Some Analytical Techniques
- Purification of Solids
- Crystallisation Method
- Fractional Crystallization
- Simple Distillation Method
- Solvent Extraction
- Chromatography Method
- Chromatography Method > Adsorption Chromatography
- Chromatography Method > Partition Chromatography
Structure of Atom
Chemical Bonding
- Concept of Chemical Bonding
- Kossel-lewis Approach to Chemical Bonding - Octet Rule
- Kossel and Lewis Approach to Chemical Bonding
- Formal Charge
- Limitations of the Octet Rule
- Valence Shell Electron Pair Repulsion Theory (VSEPR)
- Valence Bond Theory (VBT)
- Molecular Orbital Theory
- Parameters of Covalent Bond
- Dipole Moment
- Resonance
Redox Reactions
- Introduction of Redox Reactions
- Oxidation Number
- Balancing of Redox Reactions
- Redox Reaction and Electrode Potential
Modern Periodic Table
- Introduction of Periodic Table
- Structure of the Modern Periodic Table
- Periodic Table and Electronic Configuration
- Blockwise Characteristics of Elements
- Periodic Trends in Elemental Properties
Elements of Group 1 and 2
- Hydrogen
- Alkali Metals and Alkaline Earth Metals
- Some Important Compounds of Elements of S-block
Elements of Group 13, 14 and 15
- Electronic Configuration of Elements of Groups 13, 14 and 15
- Trends in Atomic and Physical Properties of Elements of Groups 13, 14 and 15
- Chemical Properties of the Elements of the Groups 13,14 and 15
- Carbon: A Versatile Element
- Allotropes of Carbon > Diamond
- Molecular Structures of Some Important Compounds of the Group 13, 14 and 15 Elements
- Chemistry of Notable Compounds of Elements of Groups 13, 14 and 15
States of Matter
- States of Matter
- Intermolecular Forces
- Characteristic Properties of Gases
- Gas Laws
- Ideal Gas Equation
- Kinetic Molecular Theory of Gases
- Deviation from Ideal Behaviour
- Liquefaction of Gases and Critical Constant
- Liquid State
Adsorption and Colloids
- Introduction of Adsorption
- Adsorption
- Types of Adsorption
- Factors Affecting Adsorption of Gases on Solids
- Adsorption Isotherms (Freundlich and Langmuir Adsorption Isotherm)
- Applications of Adsorption
- Catalysis
- Adsorption Theory of Heterogeneous Catalysis
- Colloids
Chemical Equilibrium
- Introduction of Chemical Equilibrium
- Equilibrium in Physical Processes
- Equilibrium in Chemical Processes - Dynamic Equilibrium
- Law of Mass Action and Equilibrium Constant
- Homogeneous and Heterogenous Equilibria
- Characteristics of Equilibrium Constant
- Applications of Equilibrium Constants
- Le Chaterlier's Principle and Factors Altering the Composition of Equilibrium
- Industrial Application
Nuclear Chemistry and Radioactivity
- Introduction: Nuclear Chemistry is a Branch of Physical Chemistry
- Classification of Nuclides
- Nuclear Stability
- Radioactivity
- Radioactive Decays
- Modes of Decay
- Nuclear Reactions
- Applications of Radio Isotopes
Basic Principles of Organic Chemistry
- Organic Chemistry
- Structural Representation of Organic Molecules
- Classification of Organic Compounds
- Nomenclature
- Isomerism
- Theoretical Basis of Organic Reactions
Hydrocarbons
Chemistry in Everyday Life
- Chemistry in Everyday Life
- Basics of Food Chemistry
- Compounds with Medicinal Properties
- Cleansing Agents
- Introduction
- Definition: Ideal Gas Equation
- Law: Boyle's Law
- Law: Charles' Law
- Law: Gay-Lussac's Law
- Deriving the Ideal Gas Equation
- Formula Combines GAs Law
- Example
- Real-Life Applications
- Key Points: Ideal Gas Equation
Introduction
Gases are everywhere — in the air we breathe, the tyres of our vehicles, the pressure cookers in our kitchens, and even the hot air balloons that float across the sky. Unlike solids and liquids, gases can be compressed, expanded, and heated in ways that dramatically change their behaviour.
Three measurable properties define the state of any gas:
| Property | Symbol | What It Measures | SI Unit |
|---|---|---|---|
| Pressure | P | Force exerted by gas molecules per unit area on container walls | Pascal (Pa) |
| Volume | V | Space occupied by the gas | m³ |
| Temperature | T | Measure of the average kinetic energy of molecules | Kelvin (K) |
Definition: Ideal Gas Equation
“The relation between three properties of a gas, i.e., pressure, volume and temperature, is called the ideal gas equation.”
Law: Boyle's Law — Pressure & Volume
Statement:
For a fixed mass of gas at constant temperature, the pressure is inversely proportional to the volume.
Mathematically, P ∝ \[\frac {1}{V}\] ⇒ PV = constant
Graph: P vs V (Isotherm)
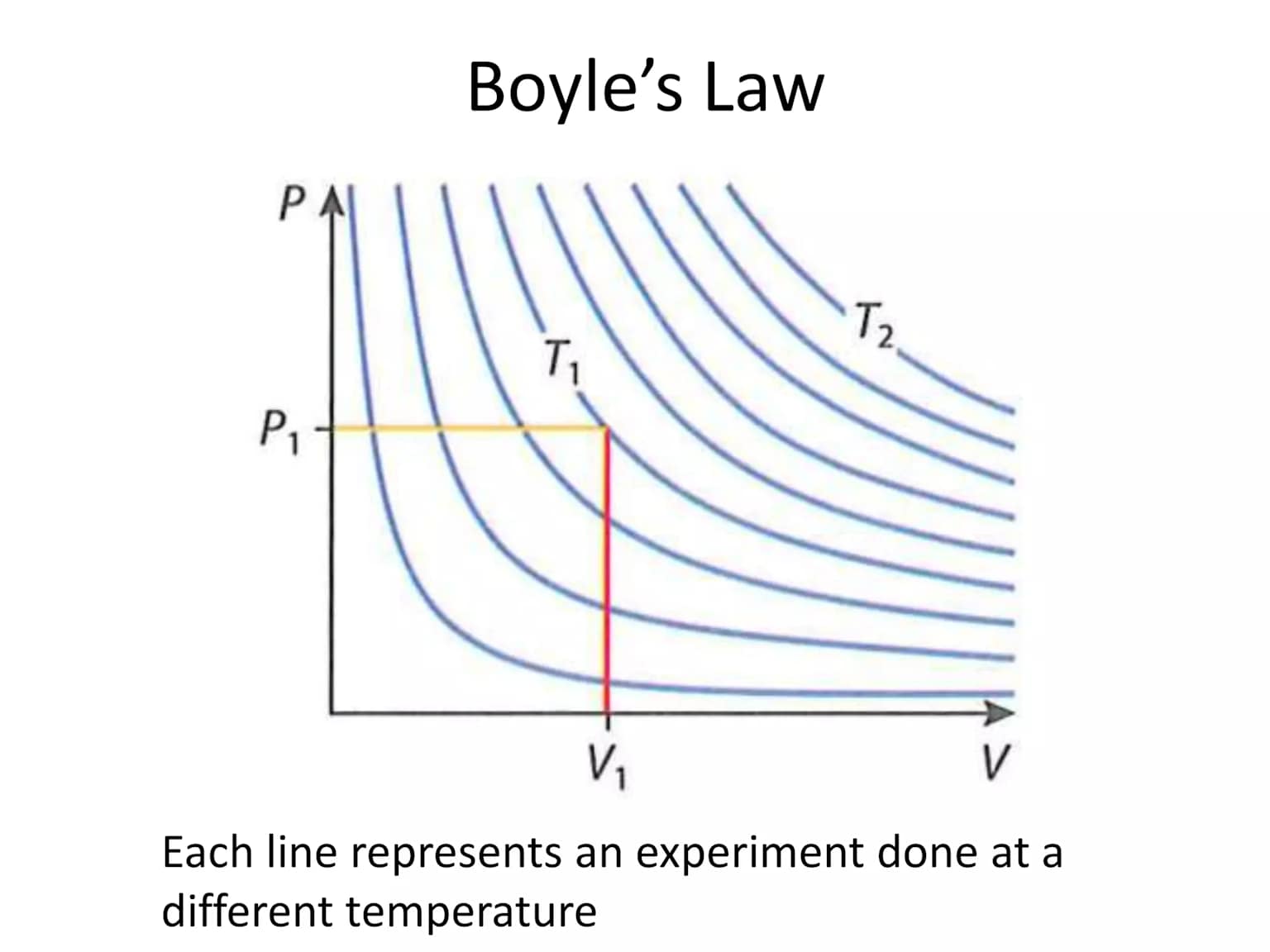
This means: squeezing a gas into a smaller space increases its pressure. Doubling the pressure halves the volume.
Law: Charles' Law — Volume & Temperature
Statement:
The volume of a fixed mass of gas is directly proportional to its absolute temperature if the pressure is kept constant.
Mathematically, V ∝ T ⇒ \[\frac {V}{T}\] = constant
Graph: V vs T (Isobar)
 A straight line through the origin when using Kelvin. All lines converge at 0 K (absolute zero).
A straight line through the origin when using Kelvin. All lines converge at 0 K (absolute zero).
Law: Gay-Lussac's Law — Pressure & Temperature
Statement:
The pressure of a fixed mass of gas is directly proportional to its absolute temperature if volume is kept constant.
Mathematically, P ∝ T ⇒ \[\frac {P}{T}\] = constant
This means: heating a gas in a sealed (rigid) container increases its pressure.
Graph: P vs T (Isochore)
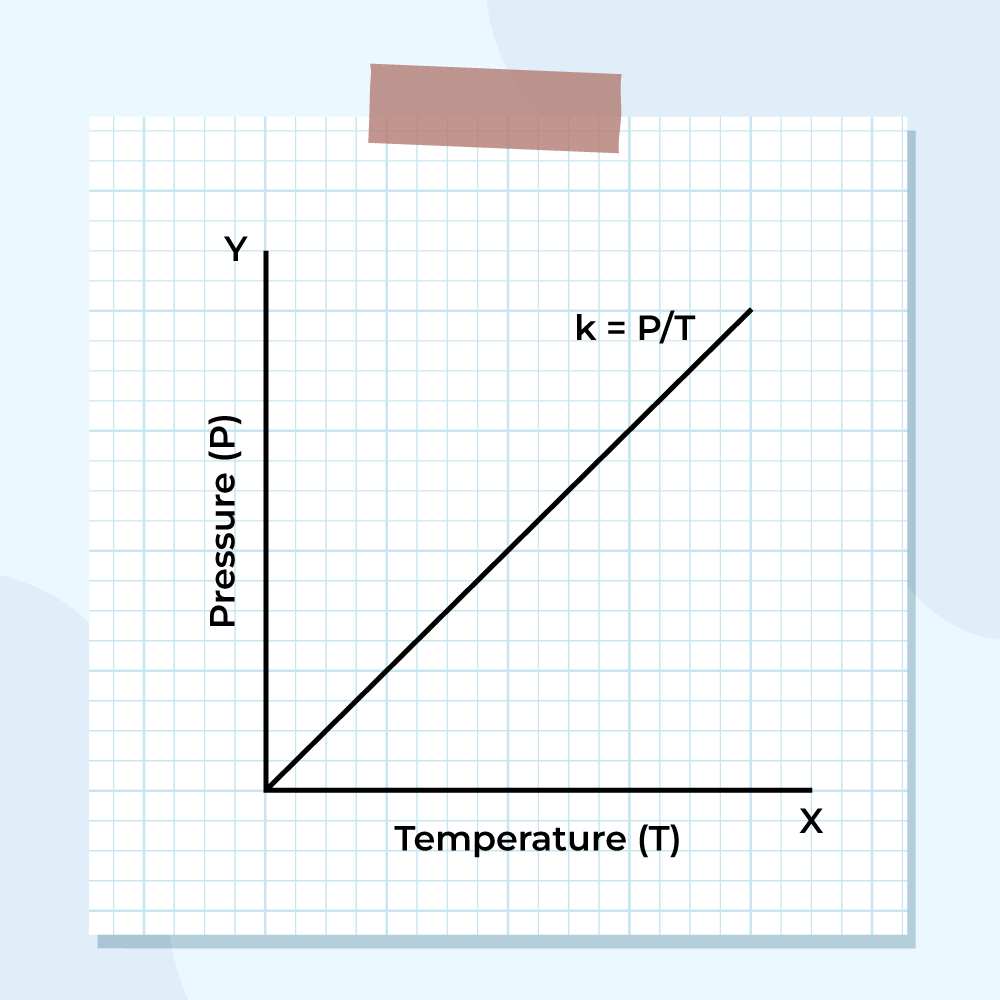
P vs T graph at constant volume — a straight line through the origin (Kelvin scale).
Deriving the Ideal Gas Equation
Step-by-Step Derivation
- From Boyle's Law (at constant T): V ∝ \[\frac {1}{P}\] ...(i)
- From Charles' Law (at constant P): V ∝ T ...(ii)
- Combining (i) and (ii): V ∝ \[\frac {T}{P}\]
- Introducing the proportionality constant R (for 1 mole): V = \[\frac {RT}{P}\]
- Rearranging: PV = RT (for 1 mole) ...(4)
- For n moles of gas: PV = nRT
where:
p = pressure (Pa)
V = volume (m³)
n = number of moles of gas
R = universal gas constant = 8.31JK-1mol-1
T = absolute temperature (K)
This is the Ideal Gas Equation — also known as the equation of state of an ideal gas.
Formula: Combined Gas Law
\[\frac{P_1V_1}{T_1}=\frac{P_2V_2}{T_2}\]
Example
Problem: The pressure reading in a thermometer at the steam point is 1.367 × 103 Pa. What is pressure reading at the triple point, knowing the linear relationship between temperature and pressure?
Step 1: Convert steam point to Kelvin: T = 273.15 + 100 = 373.15 K
Step 2: Using Gay-Lussac's Law (P ∝ T):
\[\frac {P_{triple}}{T_{triple}}\] = \[\frac {P}{T}\]
Step 3: Solve:
Ptriple = \[\frac {273.16}{373.15}\] × 1.367 × 103
Answer: Ptriple = 1.000 × 103 Pa
Real-Life Applications
- Bicycle Pump (Boyle’s Law): Decreasing air volume increases pressure, pushing air into the tyre.
- Syringe (Boyle’s Law): Pulling the plunger in increases the volume and lowers the pressure, drawing liquid into the syringe.
- Scuba Diving (Boyle’s Law): As depth increases, water pressure increases and the air volume in the lungs decreases.
- Hot Air Balloon (Charles’ Law): Heating air increases its volume, making the balloon rise.
- Bread Baking (Charles’ Law): Gas expands when heated, causing dough to rise and become fluffy.
- Car Tyres in Summer (Gay-Lussac’s Law): Higher temperature increases air pressure inside tyres.
- Pressure Cooker (Gay-Lussac’s Law): Heating increases steam pressure in a sealed pot, cooking food faster.
- Airbags (Ideal Gas Law): Rapid expansion of gas inflates airbags to protect passengers during a crash.
Key Points: Ideal Gas Equation
- An ideal gas has point-mass molecules, no intermolecular forces, and perfectly elastic collisions.
- Boyle's Law (constant T): PV = constant — pressure and volume are inversely proportional.
- Charles' Law (constant P): V/T = constant — volume and temperature are directly proportional.
- Gay-Lussac's Law (constant V): P/T = constant — pressure and temperature are directly proportional.
- The Ideal Gas Equation, PV = nRT, combines all three laws into a single universal relationship.
- The Universal Gas Constant R = 8.314 J mol⁻¹ K⁻¹ is the same for all ideal gases.
- Real gases approximate ideal behaviour at low pressure and high temperature.
- Always use absolute temperature (Kelvin) in gas law calculations. T(K) = T(°C) + 273.15
