Advertisements
Advertisements
प्रश्न
A steam engine delivers 5.4×108 J of work per minute and services 3.6 × 109 J of heat per minute from its boiler. What is the efficiency of the engine? How much heat is wasted per minute?
उत्तर १
Work done perminute, output =` 5.4 xx 10^8 J`
Heat absorbed per minute, input = `3.6 xx 10^9 J`
Efficiency, = eta = "output"/"input" = (5.4 xx 10^8)/(3.6 xx 10^9) = 0.15
`%eta = 0.15 xx 100 = 15`
Heat energy wasted /minute
= Heat energy absorbed / minute - Useful work done / minute
`= 3.6 xx 10^9 - 5.4 xx 10^8`
`= (3.6 - 0.54) xx 10^9 = 3.06 xx10^9 J`
उत्तर २
Work done by the steam engine per minute, W = 5.4 × 108 J
Heat supplied from the boiler, H = 3.6 × 109 J
Efficiency of the engine = `"Output energy"/"Inut energy"`
`:.eta = W/H = (5.4 xx 10^8)/(3.6 xx 10^9) = 0.15`
Hence, the percentage efficiency of the engine is 15 %.
Amount of heat wasted = 3.6 × 109 – 5.4 × 108
= 30.6 × 108 = 3.06 × 109 J
Therefore, the amount of heat wasted per minute is 3.06 × 109 J.
APPEARS IN
संबंधित प्रश्न
Consider the process on a system shown in figure. During the process, the work done by the system ______________ .
A gas is taken along the path AB as shown in figure. If 70 cal of heat is extracted from the gas in the process, calculate the change in the internal energy of the system.
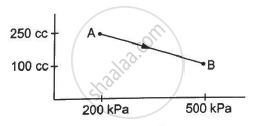
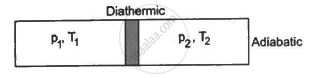
Figure shows a cylindrical tube of volume V with adiabatic walls containing an ideal gas. The internal energy of this ideal gas is given by 1.5 nRT. The tube is divided into two equal parts by a fixed diathermic wall. Initially, the pressure and the temperature are p1, T1 on the left and p2, T2 on the right. The system is left for sufficient time so that the temperature becomes equal on the two sides. (a) How much work has been done by the gas on the left part? (b) Find the final pressures on the two sides. (c) Find the final equilibrium temperature. (d) How much heat has flown from the gas on the right to the gas on the left?
Which of the following is correct, when the energy is transferred to a system from its environment?
Which of the following system freely allows the exchange of energy and matter with its environment?
What is the internal energy of the system, when the amount of heat Q is added to the system and the system does not do any work during the process?
Explain given cases related to energy transfer between the system and surrounding –
- energy transferred (Q) > 0
- energy transferred (Q) < 0
- energy transferred (Q) = 0
derive the relation between the change in internal energy (∆U), work is done (W), and heat (Q).
The internal energy of a system is ______
Explain the change in internal energy of a thermodynamic system (the gas) by heating it.
