Advertisements
Advertisements
प्रश्न
As one ascends in any homologous series, physical properties change gradually.
विकल्प
Right
Wrong
उत्तर
As one ascends in any homologous series, physical properties change gradually- Right
APPEARS IN
संबंधित प्रश्न
Write the names and formulae for the first three members of the homologous series for chloroalkanes.
The molecular formula of a homologue of butane is:
(a) C4H8
(b) C3H6
(c) C4H6
(d) C3H8
An organic compound having the molecular formula C3H6O can exist in the form of two isomers A and B having different functional groups. The isomer A is a liquid which is used as a solvent for nail polish. The isomer B is also a liquid. An aqueous solution of one of the lower homologues of B is used for preserving biological specimens in the laboratory
(a) What is compound A?
(b) Write the electron-dot structure of A.
(c) What is compound B?
(d) Write the electron-dot structure of B.
(e) Name the lower homologue of compound B which is used in preserving biological specimens.
Give the dot diagram of the first member of the alcohol.
Give the names and the structural formula of the first three members of the homologous series of alkanes.
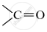 .
.
Distinguish between homologous organs and analogous organs. In which category would you place wings of a bird and wings of a bat? Justify your answer giving a suitable reason.
What is the difference in the molecular formula of any two adjacent homologues:
(i) In terms of molecular mass
(ii) In terms of number and kind of atoms per molecule?
Halo alkanes react with alkalies to produce alcohol. Give the equation for the preparation of the second member of the homologous series of alcohol. State under what condition the reaction occurs.
Complete the correlation:
Alkene : C = C :: Alkyne: _______.
While going in an increasing order there is a rise in the molecular mass of the consecutive members of the homologous series by _______.
The general molecular formula for the homologous series of alkynes is _______.
Saturated hydrocarbon : Single bond : : Unsaturated hydrocarbon : _______
Find the odd one out and give its explanation.
Complete the following table for homologous series of Alkenes.
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Ethene | C2H4 | CH2 = CH2 | 2 | 0 | -102 |
| Propene | C3H6 | CH3–CH = CH2 | 3 | 1 | -48 |
| 1-Butene | C4H8 | CH3–CH2–CH = CH2 | ______ | ______ | -6.5 |
| 1-Pentene | C5H10 | CH3–CH2–CH2–CH = CH2 | ______ | ______ | 30 |
Complete the following chart by using examples given in brackets.
(isobutylene, cyclohexane, propane, cyclohexene, cyclopentane, benzene, propyne, isobutane, propene)
| Straight chain hydrocarbons | Branched chain hydrocarbons | Cyclic hydrocarbons |
Name the following organic compound:
The first homologue whose general formula is CnH2n.
A carbon compound ‘A’ having melting point 156K and boiling point 351K, with molecular formula C2H6O is soluble in water in all proportions.
- Identify ‘A’ and draw its electron dot structure.
- Give the molecular formulae of any two homologues of ‘A’.
Define Homologous series.
