Advertisements
Advertisements
प्रश्न
Convert 0.124 torr to the standard atmosphere
उत्तर
0.124 torr to standard atmosphere:
1 atm = 760 torr
∴ 1 torr = `1/760` atm
∴ 0.124 torr = `0.124xx1/760`
= 1.632 × 10–4 atm
APPEARS IN
संबंधित प्रश्न
Give reason for the following:
Gases exert pressure in all directions.
State the following:
The absolute temperature of a gas at 7°C
Convert the following temperature from degree Celcius to kelvin.
−197° C
Convert the following pressure value into Pascals.
1 kPa
Hot air balloons float in the air because of the low density of the air inside the balloon. Explain this with the help of an appropriate gas law.

Identify the gas laws from the following diagram.
| Diagram | Gas laws |
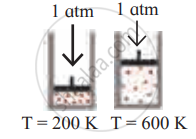 |
______________ |
Identify the gas laws from the following diagram.
| Diagram | Gas laws |
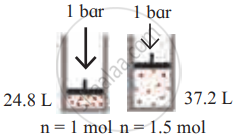 |
______________ |
Match the pairs of the following:
| Column ‘A’ | Column ‘B’ |
| a. Boyle’s law | i. at constant pressure and volume |
| b. Charles’ law | ii. at constant temperature |
| iii. at constant pressure |
Write the statement for Boyle’s law
With the help of the graph answer the following -

At constant temperature, the Graph shows the relationship between pressure and volume. Represent the relation mathematically.
Solve the following.
The volume of a given mass of a gas at 0°C is 2 dm3. Calculate the new volume of the gas at constant pressure when the temperature is increased by 10°C.
Solve the following.
At 0°C, a gas occupies 22.4 liters. How much hot must be the gas in celsius and in kelvin to reach a volume of 25.0 liters?
The temperatures at which real gases obey the ideal gas laws over a wide range of pressure is called __________.
Assertion: Critical temperature of CO2 is 304 K, it can be liquefied above 304 K.
Reason: For a given mass of gas, volume is to directly proportional to pressure at constant temperature
State Boyle's law.
Give the mathematical expression that relates gas volume and moles.
A sample of gas has a volume of 8.5 dm3 at an unknown temperature. When the sample is submerged in ice water at 0°C, its volume gets reduced to 6.37 dm3. What is its initial temperature?
Sulphur hexafluoride is a colourless, odourless gas; calculate the pressure exerted by 1.82 moles of the gas in a steel vessel of volume 5.43 dm3 at 69.5 °C, assuming ideal gas behaviour
At 25°C and 1 atm, a cylinder containing 10 L of an ideal gas is connected to the empty cylinder with a capacity of 20 L. The pressures exerted by gas m both the cylinders will be ____________.
For a given mass of an ideal gas, which of the following statements is CORRECT?
A certain sample of gas has a volume of 0.2 L at one atmosphere pressure and 273.15 K. What is the volume of gas at 273.15°C at same pressure?
A certain mass of a gas occupies a volume of 2 dm3 at STP. At what temperature the volume of gas becomes double, keeping the pressure constant?
A gas occupies a volume of 4.2 dm3 at 101 kPa pressure. What volume will gas occupy if the pressure is increased to 235 kPa keeping the temperature constant?
If 2 moles of an ideal gas at 546 K has volume of 44.8 L, then what will be it's pressure? (R = 0.082)
If 300 mL of a gas at 26.85°C is cooled to 6.85°C at constant pressure. What will be the final volume of gas?
