Advertisements
Advertisements
प्रश्न
Derive the work done in an adiabatic process.
उत्तर
Work done in an adiabatic process: Consider µ moles of an ideal gas enclosed in a cylinder having perfectly non-conducting walls and base. A frictionless and insulating piston of cross-sectional area A is fitted in the cylinder.
Let W be the work done when the system goes from the initial state (Pi, Vi, Ti) to the final state (Pf, Vf, Tf) adiabatically.
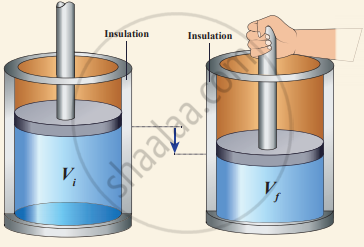
Work done in an adiabatic process
W = `int_("V"_"i")^("V"_"f") "PdV"` ..........(1)
By assuming that the adiabatic process occurs quasi-statically, at every stage the ideal gas law is valid. Under this condition, the adiabatic equation of state is PVγ = constant (or)
P = `"constant"/"V"^γ` can be substituted in the equation (1), we get
∴ Wadia = `int_("V"_"i")^("V"_"f") "constant"/"V"^γ "dV" = "constant" int_("V"_"i")^("V"_"f") "V"^-γ "dV"`
= `"constant" [("V"^(-γ + 1))/(-γ + 1)]_("V"_"i")^("V"_"f") = "constant"/(1 - γ) [1/("V"_"f"^(γ - 1)) - 1/("V"_"i"^(γ - 1))]`
= `1/(1 - γ) ["constant"/("V"_"f"^(γ - 1)) - "constant"/("V"_"i"^(γ - 1))]`
But, `"P"_"i""V"_"i"^γ = "P"_"f""V"_"f"^γ = "constant"`
∴ Wadia = `1/(1 - γ) [("P"_"f""V"_"f"^γ)/("V"_"f"^(γ - 1)) - ("P"_"i""V"_"i"^γ)/("V"_"i"^(γ - 1))]`
Wadia = `1/(1 - γ) ["P"_"f""V"_"f" - "P"_"i""V"_"i"]` .......(2)
From ideal gas law, PfVf = μRTf and PiVi = μRTi
Substituting in equation (2), we get
∴ Wadia = `(μ"R")/(γ - 1) ["T"_"i" - "T"_"f"]` ..........(3)
In adiabatic expansion, work is done by the gas. i.e., Wadia is positive. As Ti > Tf the gas cools during adiabatic expansion.
In adiabatic compression, work is done on the gas. i.e., Wadia is negative. As Ti < Tf the temperature of the gas increases during adiabatic compression.


PV diagram - Work done in the adiabatic process
To differentiate between isothermal and adiabatic curves in the PV diagram, the adiabatic curve is drawn along with the isothermal curve for Tf and Ti. Note that the adiabatic curve is steeper than an isothermal curve. This is because γ > 1 always.
APPEARS IN
संबंधित प्रश्न
What is a thermodynamic process?
An ideal gas of volume 2 L is adiabatically compressed to (1/10)th of its initial volume. Its initial pressure is 1.01 x 105 Pa, calculate the final pressure. (Given 𝛾 = 1.4)
When food is cooked in a vessel by keeping the lid closed, after some time the steam pushes the lid outward. By considering the steam as a thermodynamic system, then in the cooking process
When you exercise in the morning, by considering your body as a thermodynamic system, which of the following is true?
Derive the work done in an isothermal process.
Explain in detail an adiabatic process.
In an adiabatic expansion of the air, the volume is increased by 4%, what is the percentage change in pressure? (For air γ = 1.4)
An ideal gas is taken in a cyclic process as shown in the figure. Calculate
- work done by the gas
- work done on the gas
- Net work done in the process

For a given ideal gas 6 × 105 J heat energy is supplied and the volume of gas is increased from 4 m3 to 6 m3 at atmospheric pressure. Calculate
- the work done by the gas
- change in internal energy of the gas
- graph this process in PV and TV diagram
Assertion: Equal volumes of monatomic and polyatomic gases are adiabatically compressed separately to equal compression ratio `("P"_2/"P"_1)`. Then monatomic gas will have greater final volume.
Reason: Among ideal gases, molecules of a monatomic gas have the smallest number of degrees of freedom.
