Advertisements
Advertisements
प्रश्न
Give one equation to show the following properties of sulphuric acid:
Dehydrating property
उत्तर
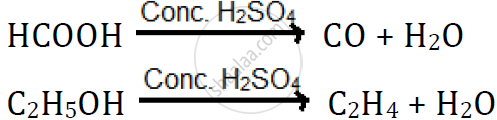
Dehydrating property of sulphuric acid:
H2SO4 has a great affinity for water, and therefore, it acts as a dehydrating agent.
APPEARS IN
संबंधित प्रश्न
Why is water not added to concentrated \[\ce{H2SO4}\] in order to dilute it?
Why is sulphuric acid known as king of chemicals and oil vitriol ?
Give examples of the use of sulphuric acid as
An oxidizing agent
Sulphuric acid can be used to prepare a number of gases in the laboratory. Write balanced equation for the reaction from which the folllowing gas are obtained, using dilute sulphuric acid as one of the reactant : Hydrogen
Write the equation for the laboratory preparation of the following salts, using sulphuric acid.
Sodium Sulphate from sodium carbonate.
Name the gas evolved in following case:
The gas produced by the action of concentrated sulphuric acid on sodium chloride.
Write the equation of the following reaction:
Sulphur is heated with concentrated sulphuric acid
State your observation for the following case :
Dry red rose petals are placed in the jar of sulphur dioxide.
State one appropriate observation of the following:
Concentrated sulphuric acid is added dropwise to a crystal of hydrated copper sulphate.
The gas evolved when dilute sulphuric acid reacts with iron sulphide.
