Advertisements
Advertisements
प्रश्न
Match the reactions given in Column I with the types of reactions given in Column II.
| Column I | Column II | |
| (i) | 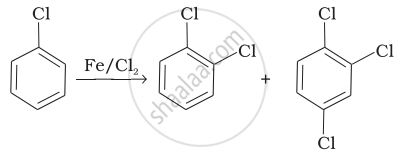 |
(a) Nucleophilic aromatic substitution |
| (ii) | \[\begin{array}{cc} \ce{CH3 - CH = CH2 + HBr -> CH3 - CH - CH3}\\ \phantom{............................}|\phantom{}\\ \phantom{.............................}\ce{Br}\phantom{} \end{array}\] |
(b) Electrophilic aromatic substitution |
| (iii) | 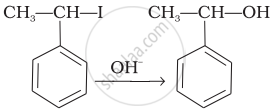 |
(c) Saytzeff elimination |
| (iv) | 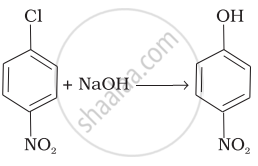 |
(d) Electrophilic addition |
| (v) | \[\begin{array}{cc} \ce{CH3 CH2 CH CH3 ->[alc.KOH] CH3 CH = CH CH3}\\ \phantom{}|\phantom{..........................}\\ \phantom{}\ce{Br}\phantom{........................} \end{array}\] |
(e) Nucleophilic substitution (SN1) |
उत्तर
| Column I | Column II | |
| (i) |  |
(b) Electrophilic aromatic substitution |
| (ii) | \[\begin{array}{cc} \ce{CH3 - CH = CH2 + HBr -> CH3 - CH - CH3}\\ \phantom{............................}|\phantom{}\\ \phantom{.............................}\ce{Br}\phantom{} \end{array}\] |
(d) Electrophilic addition |
| (iii) |  |
(e) Nucleophilic substitution (SN1) |
| (iv) |  |
(a) Nucleophilic aromatic substitution |
| (v) | \[\begin{array}{cc} \ce{CH3 CH2 CH CH3 ->[alc.KOH] CH3 CH = CH CH3}\\ \phantom{}|\phantom{..........................}\\ \phantom{}\ce{Br}\phantom{........................} \end{array}\] |
(c) Saytzeff elimination |
Explanation:
(i) In this reaction, an electrophile CF attacks on to the benzene ring and substitution takes place.
(ii) In this reaction, addition of \[\ce{HBr}\] takes place on to the doubly bonded carbons of propene in accordance with Markownikoff’s rule and electrophilic addition takes place.
(iii) In this reaction, the reactant is secondary halide. Here, halogen atom is substituted by hydroxyl ion. As it is secondary halide so it follows SN1 mechanism.
(iv) In this reaction, halogen atom is directly bonded to aromatic ring. So, it is nucleophilic aromatic substitution as OH– group has substituted halogen of given compound.
(v) It is an elimination reaction. It follows Saytzeff elimination rule.
APPEARS IN
संबंधित प्रश्न
Write the structure of an isomer of compound C4H9Br which is most reactive towards SN1 reaction
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?
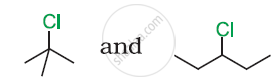
Tertiary alkyl halides are practically inert to substitution by SN2 mechanism because of ____________.
Which of the compounds will react faster in SN1 reaction with the –OH ion?
\[\ce{CH3-CH2-Cl}\] or \[\ce{C6H5-CH2-Cl}\]
Aryl halides are extremely less reactive towards nucleophilic substitution. Predict and explain the order of reactivity of the following compounds towards nucleophilic substitution:
| (I) | 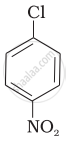 |
| (II) | 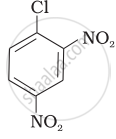 |
| (III) | 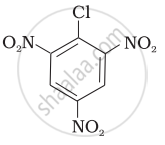 |
Why are aryl halides less reactive towards nucleophilic substitution reactions than alkyl halides? How can we enhance the reactivity of aryl halides?
Which of the following is the definition of chirality?
Which one is the correct order of nucleophilic strength (pKa) of following nucleophiles?
The number of chiral carbons present in the molecule given below is ______.
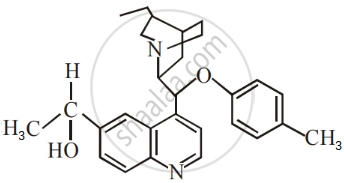
An organic compound A with the molecular formula (+) C4H9Br undergoes hydrolysis to form (+) C4H9OH. Give the structure of A and write the mechanism of the reaction.
