Advertisements
Advertisements
प्रश्न
Solve the numerical problem.
The speed of light is 3 × 108 m/s. Calculate the frequency of red light of a wavelength of 6.5 × 10−7 m.
उत्तर
Given: c = 3 × 108 m/s, λ = 6.5 × 10–7 m
To find: Frequency (ν)
Formula: c = νλ
Calculation: From formula,
ν = `"c"/λ=(3xx10^8)/(6.5xx10^-7)` = 4.6 × 1014 Hz
The frequency of red light is 4.6 × 1014 Hz.
APPEARS IN
संबंधित प्रश्न
Why are microwaves considered suitable for radar systems used in aircraft navigation?
What role dose infra-red radiation play in physical therapy?
If the earth did not have atmosphere, would its average surface temperature be higher or lower than what it is now? Explain.
To which part of the electromagnetic spectrum does a wave of frequency 5 × 1011 Hz belong?
A plane electromagnetic wave travels in vacuum along z-direction. What can you say about the directions of its electric and magnetic field vectors? If the frequency of the wave is 30 MHz, what is its wavelength?
The terminology of different parts of the electromagnetic spectrum is given in the text. Use the formula E = hv (for energy of a quantum of radiation: photon) and obtain the photon energy in units of eV for different parts of the electromagnetic spectrum. In what way are the different scales of photon energies that you obtain related to the sources of electromagnetic radiation?
Give a reason for the following:
Long-distance radio broadcasts use short-wave bands. Why?
Name the high energetic invisible electromagnetic waves which help in the study of the structure of crystals
State an additional use of the X-rays waves
Which radiation is used for satellite communication?
Give the range of wavelength of the electromagnetic waves visible to us.
What do you understand by the invisible spectrum?
State the approximate range of wavelength associated with infrared rays.
An electromagnetic wave has a frequency of 500 MHz and a wavelength of 60 cm Calculate the velocity of the wave.
The energy of a photon of a characteristic X-ray from a Coolidge tube comes from
The figure shows the intensity-wavelength relations of X-rays coming from two different Coolidge tubes. The solid curve represents the relation for the tube A in which the potential difference between the target and the filament is VA and the atomic number of the target material is ZA. These quantities are VB and ZB for the other tube. Then,
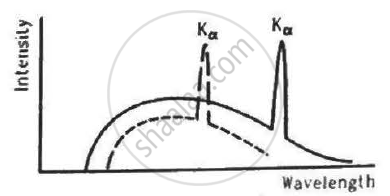
For harder X-rays,
(a) the wavelength is higher
(b) the intensity is higher
(c) the frequency is higher
(d) the photon energy is higher.
What potential difference should be applied across an X-ray tube to get X-ray of wavelength not less than 0.10 nm? What is the maximum energy of a photon of this X-ray in joule?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The Kβ X-ray of argon has a wavelength of 0.36 nm. The minimum energy needed to ionize an argon atom is 16 eV. Find the energy needed to knock out an electron from the K shell of an argon atom.
Name two sources of infrared radiation.
State two properties of infrared radiations which differ from visible light.
Give one use of electromagnetic radiations in Infrared radiation.
Answer briefly.
Name the most harmful radiation entering the Earth's atmosphere from outer space.
If the Earth did not have atmosphere, would its average surface temperature be higher or lower than what it is now? Explain.
If λv, λx and λm Am represents the wavelength of visible light, x-ray and microwaves respectively, then ______.
The electric field intensity produced by the radiations coming from 100 W bulb at a 3 m distance is E. The electric field intensity produced by the radiations coming from 50 W bulb at the same distance is ______.
Why does microwave oven heats up a food item containing water molecules most efficiently?
The half-value thickness of an absorber is defined as the thickness that will reduce exponentially the intensity of a beam of particles by a factor of 2. The half-value thickness in (µm) for lead assuming X-ray beam of wavelength 20 pm, µ = 50 cm-1 for X-rays in lead at wavelength λ = 20 pm, is ______ µm.
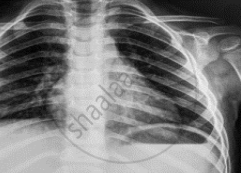
Name the electromagnetic radiation that has been used in obtaining the image below.