Advertisements
Advertisements
प्रश्न
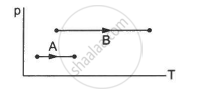
Consider two processes on a system as shown in figure.
The volumes in the initial states are the same in the two processes and the volumes in the final states are also the same. Let ∆W1 and ∆W2 be the work done by the system in the processes A and B respectively.
विकल्प
∆W1 > ∆W2
∆W1 = ∆W2
∆W1 < ∆W2
Nothing can be said about the relation between ∆W1 and ∆W2
उत्तर
∆W1 < ∆W2
Work done by the system, ∆W = P ∆ V
here,
P = Pressure in the process
∆V = Change in volume during the process
Let Vi and Vf be the volumes in the initial states and final states for processes A and B, respectively. Then,
\[\Delta W_1 = P_1 \Delta V_1 \]
\[\Delta W_2 = P_2 \Delta V_2 \]
But \[\Delta V_2 = \Delta V_1 ,.............\left[ \left( V_{f_1} - V_{i_1} \right) = \left( V_{f_2} - V_{i_2} \right) \right]\]
\[ \Rightarrow \frac{\Delta W_1}{\Delta W_2} = \frac{P_1}{P_2}\]
\[ \Rightarrow \Delta W_1 < \Delta W_2..........\left[ \because P_2 > P_1 \right]\]
APPEARS IN
संबंधित प्रश्न
Explain why Air pressure in a car tyre increases during driving.
Should the internal energy of a system necessarily increase if its temperature is increased?
A closed bottle contains some liquid. the bottle is shaken vigorously for 5 minutes. It is found that the temperature of the liquid is increased. Is heat transferred to the liquid? Is work done on the liquid? Neglect expansion on heating.
An ideal gas is pumped into a rigid container having diathermic walls so that the temperature remains constant. In a certain time interval, the pressure in the container is doubled. Is the internal energy of the contents of the container also doubled in the interval ?
Consider the process on a system shown in figure. During the process, the work done by the system ______________ .
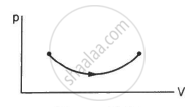
In a process on a system, the initial pressure and volume are equal to the final pressure and volume.
(a) The initial temperature must be equal to the final temperature.
(b) The initial internal energy must be equal to the final internal energy.
(c) The net heat given to the system in the process must be zero.
(d) The net work done by the system in the process must be zero.
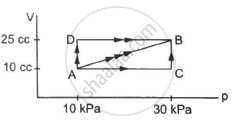
Figure shows three paths through which a gas can be taken from the state A to the state B. Calculate the work done by the gas in each of the three paths.
Which of the following is correct, when the energy is transferred to a system from its environment?
A system releases 100 kJ of heat while 80 kJ of work is done on the system. Calculate the change in internal energy.
derive the relation between the change in internal energy (∆U), work is done (W), and heat (Q).
The internal energy of a system is ______
A thermodynamic system goes from states
(i) P, V to 2P, V (ii) P, V to P, 2V
The work done in the two cases is ____________.
Two samples A and B, of a gas at the same initial temperature and pressure are compressed from volume V to V/2; A isothermally and B adiabatically. The final pressure of A will be ______.
Which of the following represents isothermal process?
Two cylinders A and B of equal capacity are connected to each other via a stopcock. A contains a gas at standard temperature and pressure. B is completely evacuated. The entire system is thermally insulated. The stopcock is suddenly opened. Answer the following:
What is the change in the temperature of the gas?
In insulated systems, the amount of external work done by the gas is proportional to:
A person of mass 60 kg wants to lose 5kg by going up and down a 10 m high stairs. Assume he burns twice as much fat while going up than coming down. If 1 kg of fat is burnt on expending 7000 kilo calories, how many times must he go up and down to reduce his weight by 5 kg?
What is heat?
