Advertisements
Advertisements
प्रश्न
In a process on a system, the initial pressure and volume are equal to the final pressure and volume.
(a) The initial temperature must be equal to the final temperature.
(b) The initial internal energy must be equal to the final internal energy.
(c) The net heat given to the system in the process must be zero.
(d) The net work done by the system in the process must be zero.
उत्तर
(a) The initial temperature must be equal to the final temperature.
(b) The initial internal energy must be equal to the final internal energy.
a) Let initial pressure, volume and temperature be P1, V1 and T1 and final pressure, volume and temperature be P2, V2 and T2. Then,
\[\frac{P_1 V_1}{T_1} = \frac{P_2 V_2}{T_2}\]
\[ \Rightarrow T_1 = T_2...........\left[ P_1 = P_2\text{ and }V_1 = V_2 \right]\]
b) Internal energy is given by ΔU = nCvΔT.
Since ΔT = 0, ΔU = 0.
c) This may not be true because the system may be isothermal and in between, heat may have been given to the system. Also, the system may have done mechanical expansion work and returned back to its original state.
d) Not necessary because an isothermal system may have done work absorbing heat from outside and coming back to the same state losing heat.
APPEARS IN
संबंधित प्रश्न
Explain why Air pressure in a car tyre increases during driving.
Should the internal energy of a system necessarily increase if its temperature is increased?
The outer surface of a cylinder containing a gas is rubbed vigorously by a polishing machine. The cylinder and its gas become warm. Is the energy transferred to the gas heat or work?
A closed bottle contains some liquid. the bottle is shaken vigorously for 5 minutes. It is found that the temperature of the liquid is increased. Is heat transferred to the liquid? Is work done on the liquid? Neglect expansion on heating.
Can work be done by a system without changing its volume?
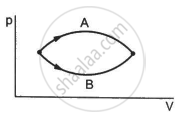
Figure shows two processes A and B on a system. Let ∆Q1 and ∆Q2 be the heat given to the system in processes A and B respectively. Then ____________ .
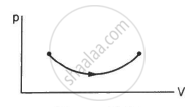
Consider the process on a system shown in figure. During the process, the work done by the system ______________ .
Consider the following two statements.
(A) If heat is added to a system, its temperature must increase.
(B) If positive work is done by a system in a thermodynamic process, its volume must increase.
A gas is contained in a metallic cylinder fitted with a piston. The piston is suddenly moved in to compress the gas and is maintained at this position. As time passes the pressure of the gas in the cylinder ______________ .
The pressure p and volume V of an ideal gas both increase in a process.
(a) Such a process is not possible.
(b) The work done by the system is positive.
(c) The temperature of the system must increase.
(d) Heat supplied to the gas is equal to the change in internal energy.
Define heat.
What is the internal energy of the system, when the amount of heat Q is added to the system and the system does not do any work during the process?
Explain given cases related to energy transfer between the system and surrounding –
- energy transferred (Q) > 0
- energy transferred (Q) < 0
- energy transferred (Q) = 0
An ideal gas is compressed at a constant temperature. Its internal energy will ____________.
Figure shows the P-V diagram of an ideal gas undergoing a change of state from A to B. Four different parts I, II, III and IV as shown in the figure may lead to the same change of state.

- Change in internal energy is same in IV and III cases, but not in I and II.
- Change in internal energy is same in all the four cases.
- Work done is maximum in case I
- Work done is minimum in case II.
A gas is compressed at a constant pressure of 50 N/m2 from a volume of 10 m3 to a volume of 4 m3. Energy of 100 J is then added to the gas by heating. Its internal energy is ______.
The internal energy of one mole of argon at 300 K is ______. (R = 8.314 J/mol.K)
A system releases 125 kJ of heat while 104 kJ work is done on the system. Calculate the change in internal energy.
Explain the change in internal energy of a thermodynamic system (the gas) by heating it.
