Advertisements
Advertisements
प्रश्न
A system can be taken from the initial state p1, V1 to the final state p2, V2 by two different methods. Let ∆Q and ∆W represent the heat given to the system and the work done by the system. Which of the following must be the same in both the methods?
विकल्प
∆Q
∆W
∆Q + ∆W
∆Q − ∆W
उत्तर
∆Q − ∆W
A system is taken from an initial state to the final state by two different methods. So, work done and heat supplied in both the cases will be different as they depend on the path followed. On the other hand, internal energy of the system (U) is a state function, i.e. it only depends on the final and initial state of the process. They are the same in the above two methods.
Using the first law of thermodynamics, we get
∆Q = ∆U - ∆W
⇒ ∆U = ∆Q - ∆W
Here, ∆U is the change in internal energy, ∆Q is the heat given to the system and ∆W is the work done by the system.
APPEARS IN
संबंधित प्रश्न
Write the mathematical expression of the First Law of Thermodynamics for Isothermal Process
Write the mathematical expression of the First Law of Thermodynamics for the Isobaric process.
Answer the following in one or two sentences.
State the first law of thermodynamics.
Define an isolated system.
A sample of gas absorbs 4000 kJ of heat and surrounding does 2000 J of work on sample. What is the value of ∆U?
A gas performs 0.320 kJ work on surrounding and absorbs 120 J of heat from the surrounding. Hence, change in internal energy is ______.
The isothermal bulk modulus of a perfect gas at pressure P is numerically equal to ____________.
Three copper blocks of masses M1, M2 and M3 kg respectively are brought into thermal contact till they reach equilibrium. Before contact, they were at T1, T2, T3 (T1 > T2 > T3). Assuming there is no heat loss to the surroundings, the equilibrium temprature T is (s is specific heat of copper)
Consider one mole of perfect gas in a cylinder of unit cross section with a piston attached (figure). A spring (spring constant k) is attached (unstretched length L) to the piston and to the bottom of the cylinder. Initially the spring is unstretched and the gas is in equilibrium. A certain amount of heat Q is supplied to the gas causing an increase of volume from V0 to V1.

- What is the initial pressure of the system?
- What is the final pressure of the system?
- Using the first law of thermodynamics, write down a relation between Q, Pa, V, Vo and k.
The first law of thermodynamics is concerned with the conservation of ______.
A system is taken through a cyclic process represented by a circle as shown. The heat absorbed by the system is ______.
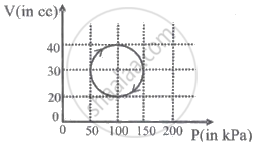
200g water is heated from 40°C to 60°C. Ignoring the slight expansion of water, the change in its internal energy is close to ______.
(Given specific heat of water = 4184 J/kgK)
The first law of thermodynamics for isothermal process is ______.
One mole of an ideal gas is allowed to expand reversibly and adiabatically from a temperature of 27°C. If the work done during the process is 3 kJ, the final temperature will be equal to ______.
(Cv = 20 JK−1)
The V cc volume of gas having `γ = 5/2` is suddenly compressed to `(V/4)` cc. The initial pressure of the gas is p. The final pressure of the gas will be ______.
What work will be done, when 3 moles of an ideal gas are compressed to half the initial volume at a constant temperature of 300 K?
A given system undergoes a change in which the work done by the system equals the decrease in its internal energy. The system must have undergone an ______.
In an adiabatic expansion of 2 moles of a gas, the initial pressure was 1.013 × 105 Pa, the initial volume was 22.4 L, the final pressure was 3.191 × 104 Pa and the final volume was 44.8 L. Find the work done by the gas on its surroundings. Taken `γ = 5/3`.
Show that the heat absorbed at constant pressure is equal to the change in enthalpy of the system.
Calculate work done when 2 moles of ideal gas expands by 5 dm3 isothermally at pressure 1.2 bar.
