Advertisements
Advertisements
प्रश्न
Two molecules of a gas have speeds of 9 × 10 6 ms−1 and 1 × 106 ms−1, respectively. What is the root mean square speed of these molecules?
उत्तर
For n-molecules, we know that
`v_(rms) = sqrt((v_1^2 + v_2^2 + v_3^2 + ...... + v_n^2)/n` .....`[(v_(rms) = "root mean"),("square velocity")]`
Where v1, v2, v3 ....... vn are individual velocities of n-molecules of the gas.
For two molecules,
`v_(rms) = sqrt((v_1^2 + v_2^2)/2` ......[v1, v2, v3 ....... vn are individual velocity]
Given, `v_1 = 9 xx 10^6` m/s
And `v_2 = 1 xx 10^6` m/s
∴ `v_(rms) = sqrt(((9 xx 10^6)^2 + (1 xx 10^6)^2)/2`
= `sqrt((81 xx 10^12 + 1 xx 10^12)/2`
= `sqrt(((81 + 1) xx 10^12)/2`
= `sqrt((82 xx 10^12)/2`
= `sqrt(41) xx 10^6` m/s
APPEARS IN
संबंधित प्रश्न
The temperature and pressure at Simla are 15.0°C and 72.0 cm of mercury and at Kalka these are 35.0°C and 76.0 cm of mercury. Find the ratio of air density at Kalka to the air density at Simla.
Use R=8.314J K-1 mol-1
At what temperature the mean speed of the molecules of hydrogen gas equals the escape speed from the earth?
Use R = 8.314 JK-1 mol-1
Air is pumped into the tubes of a cycle rickshaw at a pressure of 2 atm. The volume of each tube at this pressure is 0.002 m3. One of the tubes gets punctured and the volume of the tube reduces to 0.0005 m3. How many moles of air have leaked out? Assume that the temperature remains constant at 300 K and that the air behaves as an ideal gas.
Use R = 8.3 J K-1 mol-1
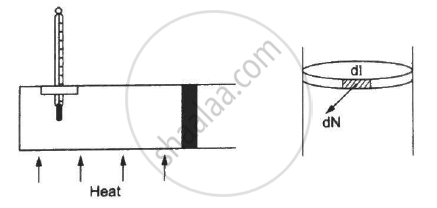
Figure shows a cylindrical tube of radius 5 cm and length 20 cm. It is closed by a tight-fitting cork. The friction coefficient between the cork and the tube is 0.20. The tube contains an ideal gas at a pressure of 1 atm and a temperature of 300 K. The tube is slowly heated and it is found that the cork pops out when the temperature reaches 600 K. Let dN denote the magnitude of the normal contact force exerted by a small length dlof the cork along the periphery (see the figure). Assuming that the temperature of the gas is uniform at any instant, calculate `(dN)/(dt)`.
In an ideal gas, the molecules possess
On what, the values of absorption coefficient, reflection coefficient, and transmission coefficient depend, in addition to the material of the object on which the radiation is an incident?
A metal cube of length 4 cm radiates heat at the rate of 10 J/s. Find its emissive power at a given temperature.
A molecule consists of two atoms each of mass 'm' and separated by a distance 'd'. At room temperature the average rotational kinetic energy is 'E', then its angular frequency is ______.
Two gases A and B are at absolute temperatures of 360 K and 420 K, respectively. The ratio of the average kinetic energy of the molecules of gas B to that of gas A is ______.
When a particle oscillates simple harmonically, its kinetic energy varies periodically. If frequency of the particle is n, then the frequency of the kinetic energy is ______.
