Advertisements
Advertisements
प्रश्न
Write the chemical equation for the preparation of 4 KMnO from 2. MnO Why does purple colour of acidified permanganate solution decolourise when it oxidises Fe2- to Fe3+ ?
उत्तर
Chemical equations for the preparation of KMnO4 from MnO2
2MnO2 + 4KOH + O2 → 2K2MnO4 + 2H2O
Purple colour of acidified permanganate solution decolourise when it oxidises Fe2- + Fe3+
In acidic medium, Reduction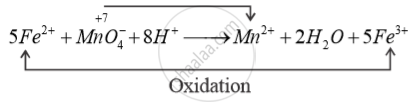
KMnO4 is behaving as oxidising Agent.
So, purple colour of acidified 4 KMnO solutions decolourises.
APPEARS IN
संबंधित प्रश्न
Complete the following equations : 2 MnO2 + 4 KOH + O2 →
Complete the following equations : MnO4- + 4H+ + 3e- →
Give an example and suggest a reason for the following feature of the transition metal chemistry:
The lowest oxide of transition metal is basic, the highest is amphoteric/acidic.
Complete the following equations:
In the form of dichromate,
(i)
(ii)
(iii) Higher oxidation states of heavier members of group-6 of transition series are more stable.
(iv) Lower oxidation states of heavier members of group-6 of transition series are more stable.
What is the effect of pH on dichromate ion solution?
Complete the reaction mentioning all the products formed:
Indicate the steps in the preparation of
Indicate the steps in the preparation of
Describe the preparation of potassium permanganate. How does the acidified permanganate solution react with SO2? Write the ionic equation for the reaction.
